D5.2 Bond Polarity
If the two atoms that form a covalent bond are identical, as in H2 or Cl2, then the electrons in the bond must be shared equally between the two atoms. In a pure covalent bond, shared electrons have an equal probability of being near each nucleus.
On the other hand, if the two atoms are different, they may have different attractions for the shared electrons. When the bonding electrons are attracted by one atom more than the other atom the bond is called a polar covalent bond. For example, in HCl, the Cl atom attracts the bonding pair of electrons more than the H atom, and electron density of the H–Cl bond is shifted towards the Cl atom. Quantum mechanics calculations show that the Cl atom, which has 17 protons, has electron density equivalent to 17.28 electrons and therefore a partial negative charge, δ– = −0.28. The hydrogen atom has a partial positive charge, δ+ = +0.28.
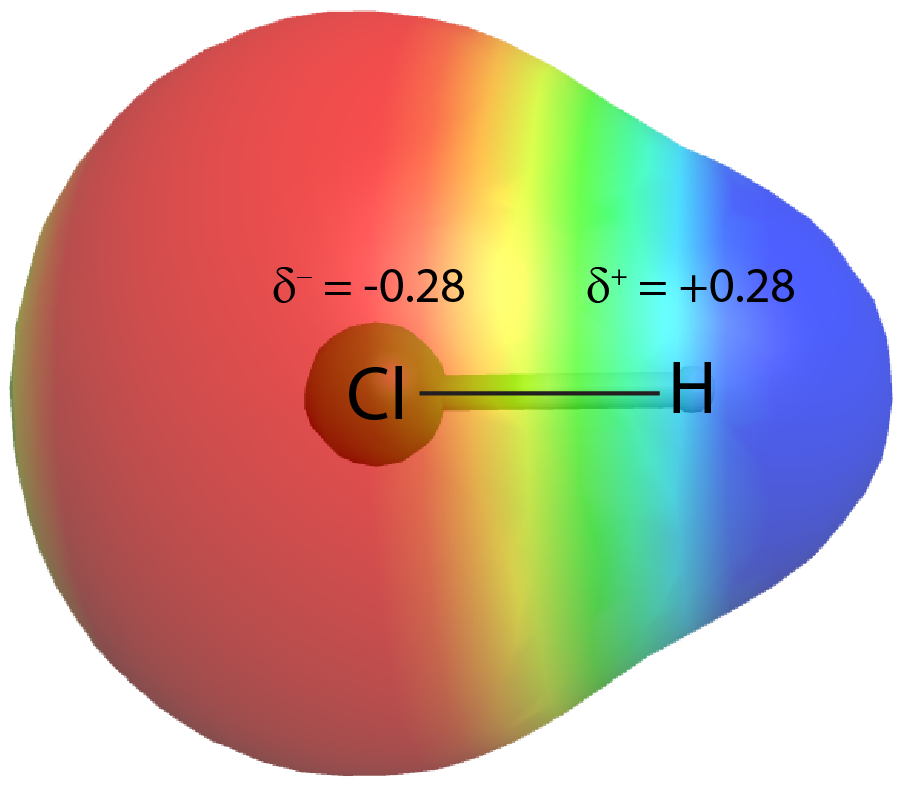
This unequal distribution of electron density on two bonded atoms produces a bond dipole moment, the magnitude of which is represented by µ (Greek letter mu):
where Q is the magnitude of the partial charges (for HCl this is 0.28 times the charge of an electron) and r is the distance between the charges (the bond length). Bond dipole moments are measured in units of debyes (D); 1 D = 3.336 × 10-30 Coulomb·meter.
The bond dipole moment (![]() ) has both direction and magnitude and can be represented as a vector (see Figure: Bond Dipole Moment). A dipole vector is drawn as an arrow, with the arrowhead pointing to the partially negative end, and a small + sign on the partially positive end. The length of the arrow is proportional to the magnitude of the dipole moment.
) has both direction and magnitude and can be represented as a vector (see Figure: Bond Dipole Moment). A dipole vector is drawn as an arrow, with the arrowhead pointing to the partially negative end, and a small + sign on the partially positive end. The length of the arrow is proportional to the magnitude of the dipole moment.
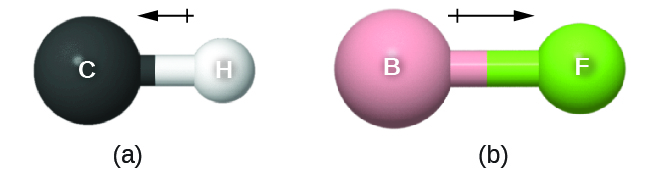
Why is the direction of the arrow different from what I learned in physics?
In physics, by convention, a dipole vector points toward the positive end of the dipole.
In chemistry, electrons (with negative charge) are more important because the position of electron density relative to atomic nuclei defines chemical bonds. Thus, in chemistry, dipoles emphasize the direction of greater electron density and the dipole arrow points toward the negative end of the dipole.
The polarity of a covalent bond can be estimated by the difference between the electronegativities of the bonded atoms. In a bond, the more electronegative atom is the one with the δ− charge. The greater the difference in electronegativity between two bonded atoms, the larger the shift of electron density in the bond towards the more electronegative atom. Greater electronegativity difference, Δ(EN), gives larger partial charges on the atoms.
Electronegativity and Bond Type
The difference in electronegativity, Δ(EN), of two bonded atoms provides a rough estimate of polarity of the bond, and thus of the bond type. When Δ(EN) is very small (or zero), the bond is covalent and nonpolar. When Δ(EN) is large, the bond is polar covalent or ionic. (In a pair of ions, such as Na+Cl−, there is nearly complete transfer of valence electrons from one atom to another to produce a positive ion and a negative ion. The Na+ and Cl− ions form a dipole with δ+ approximately equal to +1 and δ− approximately −1.)
Δ(EN) spans a continuous scale and serves as a general guide; there is no definitive cutoff that defines a bond type. For example, HF has Δ(EN) = 1.8 and is considered a polar covalent molecule. On the other hand, NaI has a Δ(EN) of 1.7 but is considered an ionic compound. When estimating the covalent or ionic character of a bond, you should also take into account the types of atoms involved and their relative positions in the periodic table. Bonds between two nonmetals are usually described as covalent; bonding between a metal and a nonmetal is often ionic.
Some compounds contain both covalent and ionic bonds. For example, potassium nitrate, KNO3, contains the K+ cation and the polyatomic NO3− anion, which has covalent bonds between N and O.
Exercise: Polarity and Electronegativity Difference
Exercise: Bond Polarity and Electronegativity
Molecular Dipoles
Applying Core Ideas: Comparing Propane and Dimethyl Ether
The additional IMF alluded to in the Applying Core Ideas box is called dipole-dipole attraction, which is an attractive electrostatic force between polar molecules. The attractive force arises when the positive end of one molecular dipole interacts with the negative end of another molecular dipole.
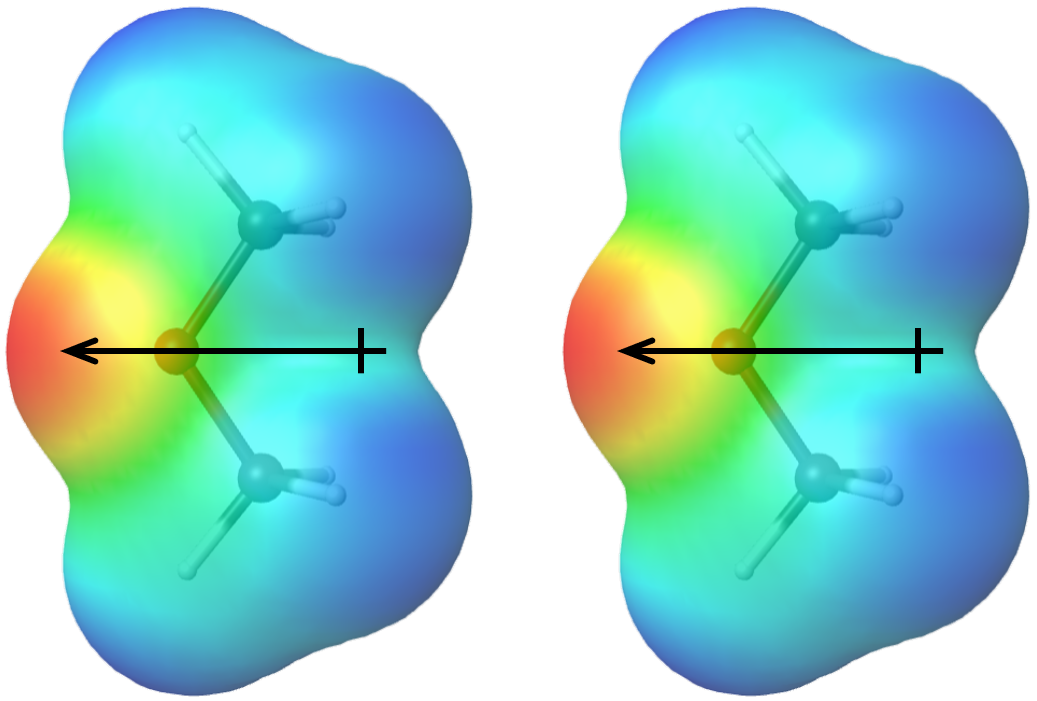
The more polar a molecule is (that is, the larger its molecular dipole moment is), the stronger the dipole-dipole attractions are between molecules of that substance. Molecular polarity depends both on the sizes of the bond dipoles (that is, on electronegativity differences between pairs of bonded atoms) and the shape of the molecule. Physical properties of a substance are influenced by all IMFs between molecules of the substance, so it is important to consider both LDFs and dipole-dipole attractions when predicting properties such as boiling points.
How do we know whether a molecule has a dipole moment? At the beginning of this section, we described polar covalent bonds—bonds in which there is an unequal distribution of electron density over two bonded atoms and hence a bond dipole moment. The sum of all bond dipole moments in a molecule gives a molecular dipole moment. Molecules that have a molecular dipole moment are called polar molecules; molecules that have a zero (or near zero) molecular dipole moment are called nonpolar molecules.
To predict whether a molecule is polar, first determine whether there are polar bonds by comparing electronegativities of each pair of bonded atoms. If electronegativity differences are small or zero, there are no polar bonds and the molecule must be nonpolar. If there are polar bonds, the molecule might be polar, but it is also possible that the bond dipoles might cancel.
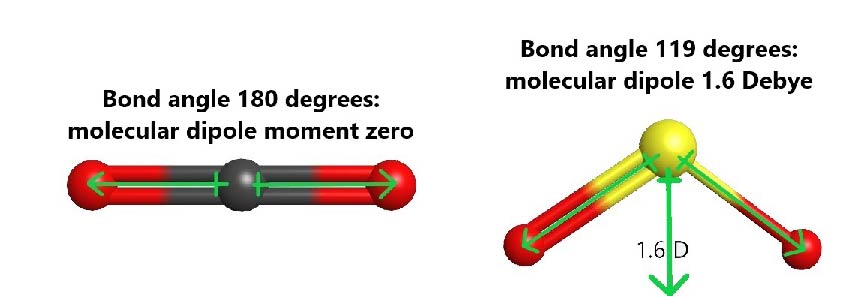
For example, both carbon dioxide (CO2) and sulfur dioxide (SO2) have polar bonds, but only SO2 is polar. In CO2, the central carbon has two σ bonds, it is sp hybridized, and therefore the molecule is linear. The bond dipole of each C=O bond is large (ΔEN = 3.5 − 2.6 = 0.9) and equal in magnitude. However, the two C=O bonds are pointing in exact opposite directions, and result in a molecular dipole moment of zero. In SO2, where the central S atom has two σ bonds and one lone pair, the S atom is sp2 hybridized and the molecule is bent. The sulfur-oxygen bond dipoles are also large (ΔEN = 3.5 − 2.6 = 0.9), but they are situated at an angle and the resultant molecular dipole is not zero.
Bond dipoles behave as vectors, so if you are familiar with vector addition you can predict when bond dipoles cancel and when they do not. Another way to predict is this: molecules with all terminal atoms the same and no lone pairs on the central atom are nonpolar because of cancellation of bond dipoles. (In the case of a molecule with an odd number of electrons, a single electron on the central atom counts as a lone pair.) For multicentered molecules, predicting molecular dipoles is trickier. Generally, if atoms have similar electronegativities, then bond dipoles are weak and the molecular dipole moment is small. For example, because C and H have similar electronegativity, C-H bonds have small bond polarity, and hydrocarbon molecules are nonpolar. The dipole moment of propane, for example, is less than 0.1 D—essentially negligible.
Exercise: Predicting Molecular Polarity
Exercise: Predicting Boiling Points
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂 (Note that we cannot answer questions via the google form. If you have a question, please post it on Piazza.)

