D8.3 Hess’s Law
The standard enthalpy change for a particular reaction is the difference between the standard enthalpy of the products and the standard enthalpy of the reactants. That difference is same regardless of whether the reaction is carried out directly or in two or more steps. This fact allows us to calculate the enthalpy change for a chemical reaction from other experimentally determined enthalpy changes. This type of calculation usually involves Hess’s law: if a process can be written as the sum of several stepwise processes, the enthalpy change of the total process equals the sum of the enthalpy changes of the various steps.
For example, we can think of the following reaction occurring in a single step:
or in a two-step process, one in which carbon is partially oxidized to CO and a second in which CO is oxidized further to the overall product CO2:
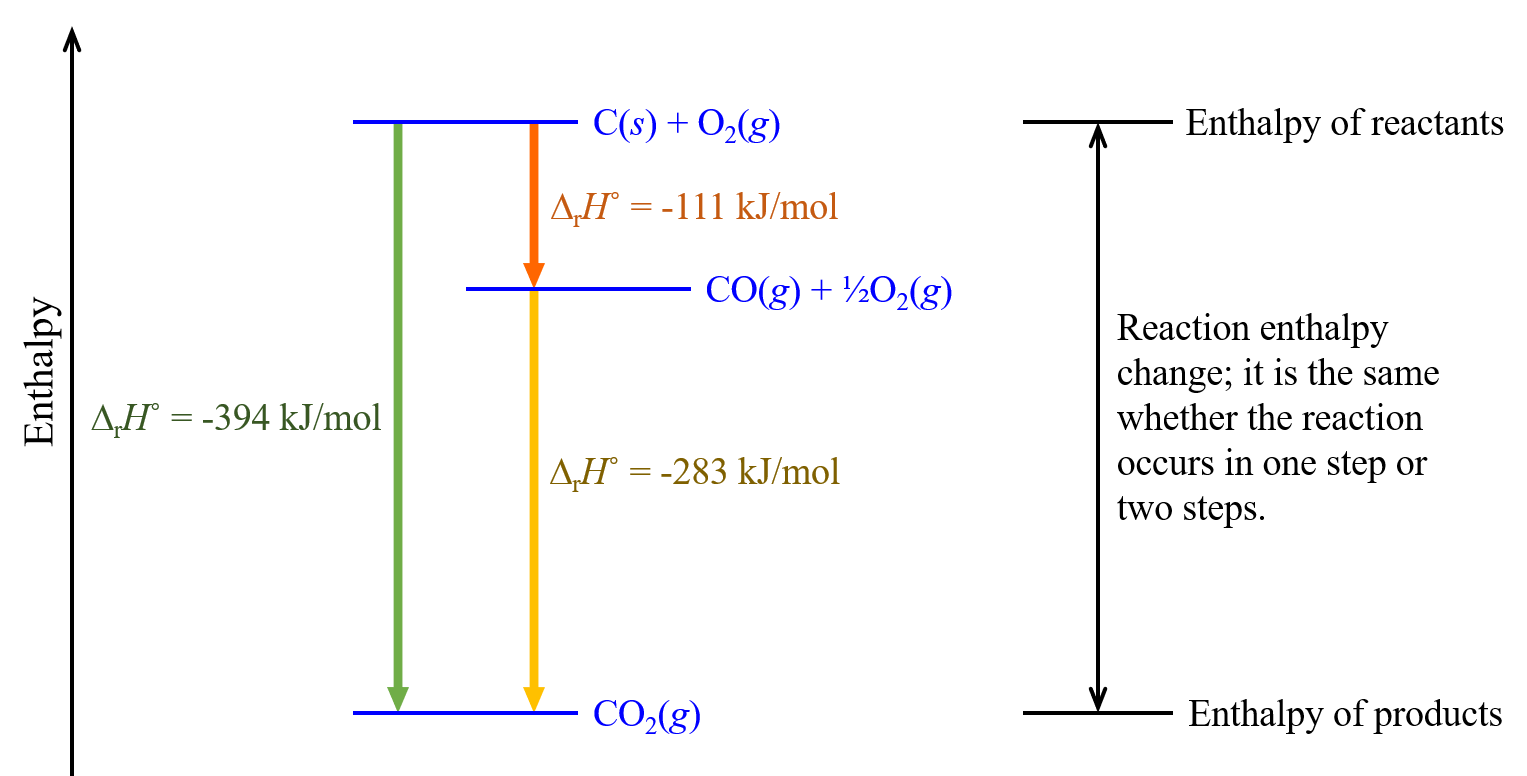
| step 1: | C(s) + ½O2(g) | ⟶ | CO(g) | ΔrH° = -111 kJ/mol |
| step 2: | CO(g) + ½O2(g) | ⟶ | CO2(g) | ΔrH° = -283 kJ/mol |
According to Hess’s law, the ΔrH° of the one-step reaction equals the sum of the ΔrH° of the two steps in the two-step reaction:
| step 1: | C(s) + ½O2(g) | ⟶ | CO(g) | ΔrH° = -111 kJ/mol |
| step 2: | CO(g) + ½O2(g) | ⟶ | CO2(g) | ΔrH° = -283 kJ/mol |
| Sum: | C(s) + O2(g) + |
⟶ | ΔrH° = -394 kJ/mol |
Note that, because it appears on both sides of the equation arrow, CO(g) is cancelled. To think about this pictorially, consider this figure.
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂 (Note that we cannot answer questions via the google form. If you have a question, please post it on Piazza.)

