D34.4 pH and pOH
The concentrations of H3O+ and OH– in a solution are important for the solution’s acid-base properties and often affect the chemical behaviors of other solutes. A solution is neutral if its [H3O+] = [OH–]; acidic if its [H3O+] > [OH–]; and basic if its [H3O+] < [OH–].
A common means of expressing values that span many orders of magnitude is to use a logarithmic scale. One such scale is based on the p-function:
where “X” is the quantity of interest and “log” is the base-10 logarithm. The pH of a solution is therefore defined as:
The reason for dividing by the units “mol/L” (M) is that [H3O+] has units of mol/L and taking the logarithm of a unit makes no sense. From here on we will assume that you are aware that only the numeric value of a concentration (or other quantity) needs to be used as the argument of a logarithm and we will not explicitly divide by the units.
If a pH value is known, the concentration of hydronium ions can be calculated:
Here we assume that you know that units are required for the concentration obtained from this equation and the units are mol/L.
The hydroxide ion concentration may be similarly expressed as pOH:
Finally, the relation between pH and pOH can be derived from the Kw expression:
| Kw | = | [H3O+][OH–] |
| -log(Kw) | = | -log([H3O+][OH–]) |
| pKw | = | -log([H3O+]) + (-log([OH–])) |
| pKw | = | pH + pOH |
At 25 °C:
Therefore, at this temperature:
| Classification | Relative Ion Concentrations | pH at 25 °C |
|---|---|---|
| acidic | [H3O+] > [OH−] | < 7 |
| neutral | [H3O+] = [OH−] | 7 |
| basic | [H3O+] < [OH−] | > 7 |
Because Kw is temperature dependent, the correlations between pH values and the acidic/neutral/basic adjectives varies at different temperatures. For example, [H3O+] in pure water at 80 °C is 4.9 × 10−7 M, which corresponds to pH and pOH values of:
pOH = -log[OH–] = -log(4.9 × 10−7) = 6.31
At this temperature, neutral solutions have pH = pOH = 6.31, acidic solutions have pH < 6.31 and pOH > 6.31, and basic solutions have pH > 6.31 and pOH < 6.31. This distinction can be important when studying certain processes that occur at temperatures other than 25 °C, such as acid-base reactions in the human body where temperatures are typically 37 °C.
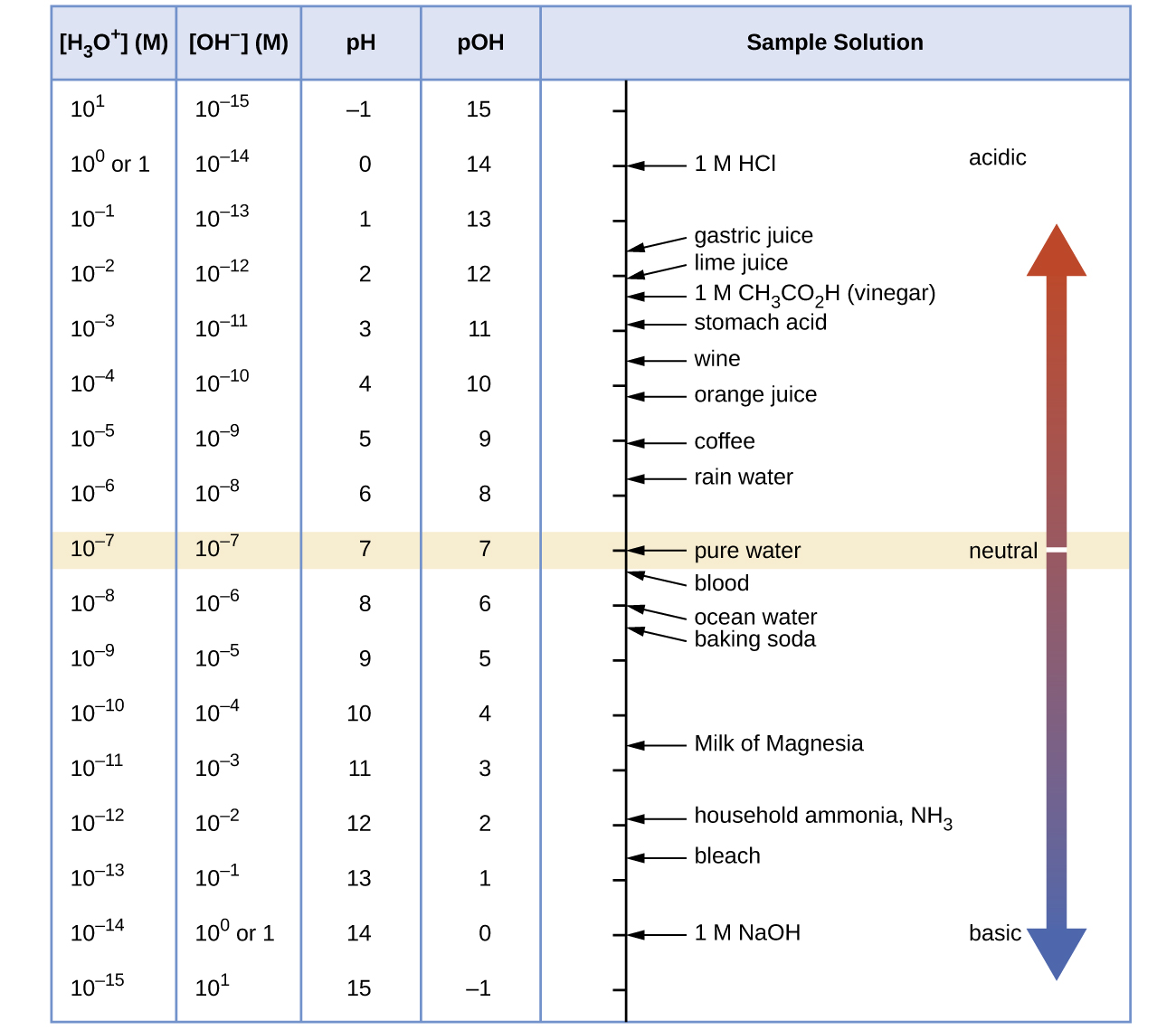
Unless otherwise noted, references to pH values are presumed to be those at 25 °C. The table below shows the relationships among [H3O+], [OH−], pH, and pOH, and gives these values for some common substances.
Activity: pH and Relative Strengths of Acids
Experiments show that the pH of a 0.010 M HCN aqueous solution is higher than the pH of a 0.010 M CH3COOH aqueous solution. Based on this observation, what can you deduce about the relative acidity of HCN (prussic acid) and CH3COOH (acetic acid)? Explain your answer.
Write in your notebook, then left-click here for an explanation.
The pH is lower for acetic acid. Based on this observation, acetic acid is a stronger acid than HCN.
The concentrations of the two acids are the same. Stronger acids are better H+ donors than weaker acids, so the same concentration of a stronger acid gives a greater concentration of H3O+, leading to a lower pH value.
Exercise: pH of Aqueous Solutions
The acidity of a solution is typically determined by measuring its pH. The pOH of a solution is not usually measured, but it is easily calculated from an experimentally determined pH value. The pH of a solution can be directly measured using a pH meter or visually estimated using colored indicators.


Activity: Determining Ka
Activity: Using Ka to Calculate Concentrations
The percent ionization of a weak acid is another measure of the strength of an acid, HA:
A stronger acid, with a larger Ka, has higher percent ionization than a weaker acid (for the same concentration). The percent ionization for a solution of a weak acid is concentration dependent: it increases with decreasing acid concentration.
Exercise 4: Percent Ionization
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂 (Note that we cannot answer questions via the google form. If you have a question, please post it on Piazza.)

