Each pre-class Podia question is based on the pre-class material and working through the pre-class material will help you formulate your response. Consider the problem and write down/draw out your solution in your class notebook. These questions are designed to hone your skills so that you can analyze and solve mastery problems you will encounter throughout the course.
Two days before the next whole-class session, the pre-class Podia question will become visible in Podia, where you can click on the prompt and submit your answer.
Submissions for pre-class Podia activities are due 1 hour before the start of your whole-class. Be sure to submit your response before the due time.
Day 35 Pre-Class Podia Activity
Alcohols (R-OH) are generally a lot less acidic (pKa ~16) than carboxylic acids (R-COOH) (pKa ~5). This can be explained by important resonance structures which delocalize the negative charge in the carboxylate ion. 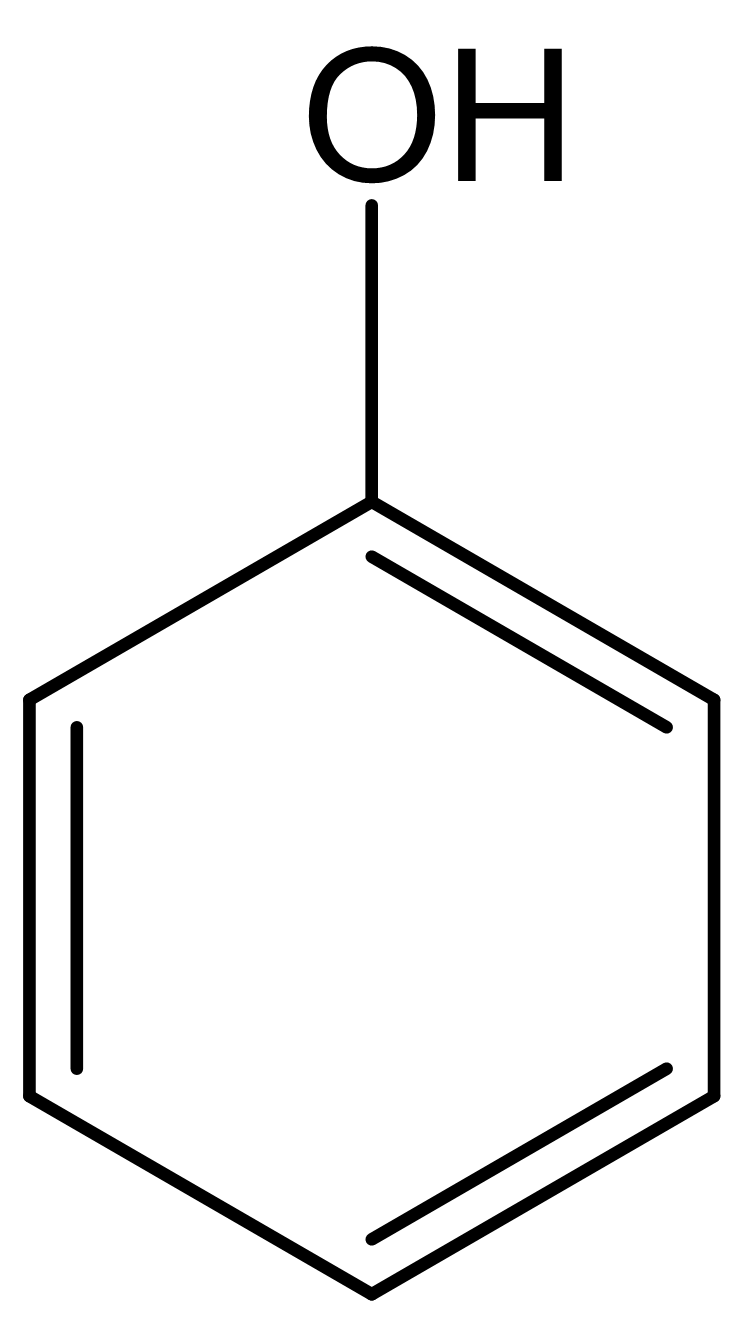
Phenol (shown right) is a lot more acidic (pKa = 10) than most alcohols. Draw important structures for the phenolate ion and use an argument based on free energy differences to describe how they explain the increased acidity of phenol compared to other alcohols.
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂 (Note that we cannot answer questions via the google form. If you have a question, please post it on Piazza.)

