D7.3 Lewis Structures for Covalent Molecules
Molecular orbitals for molecules with three or more atoms are complicated and hard to draw. Thus, although MOs would convey a more descriptive and accurate picture of electron distribution within a molecule, chemists often rely on simpler diagrams to depict the covalent bonding. It will aid your understanding of chemistry if you can connect these simpler diagrams mentally with the more complete picture given by MOs.
The most commonly used hand-drawn depiction is the Lewis structure: a diagram that represents atomic nuclei and core electrons by chemical symbols and valence electrons as dots or lines. A Lewis structure is built by combining Lewis diagrams of the constituent atoms.
Activity: Lewis Diagrams
Consider each element listed below. In your course notebook write the electron configuration for an atom of each element, determine the number of valence electrons, and draw a Lewis diagram. How are the Lewis diagrams related to the position of each element in the periodic table?
N C S As O Br F Si H
After writing and drawing in your notebook, left-click here for a comparison.
Lewis diagrams for atoms in the same vertical column (group) in the periodic table have the same number of dots. The number of dots (number of valence electrons) equals the group number in the periodic table or the group number minus 10. For example As is in group 15 (VA) so the number of valence electrons is 5.
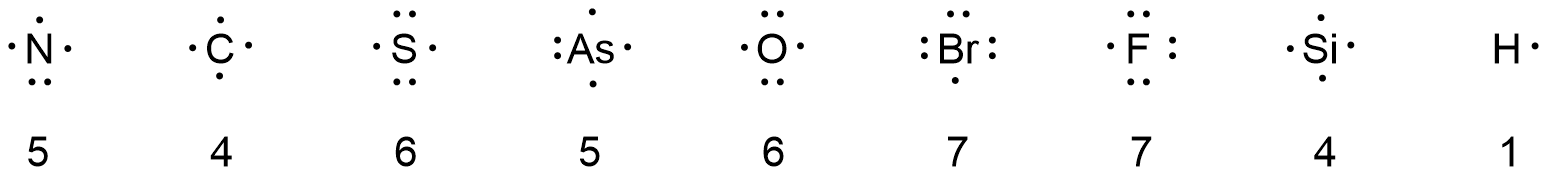
The Lewis diagram for each atom is shown above with the number of valence electrons below the atom symbol.
In Lewis structures, a single covalent bond is drawn as a pair of electron dots shared between two adjacent atoms, a bond pair. Valence electrons that are not in a bond are shown as pairs of dots associated with individual atoms, lone pairs. For example:
![]()
In the Cl2 molecule, each Cl atom has three lone pairs and the two Cl atoms share one bond pair. Hence, each Cl atom in Cl2 has formed an octet (is surrounded by eight valence electrons).
For simplicity and clarity, a bond pair is typically represented by a line instead of a pair of dots:

Activity: Lewis Structure and Electron Sharing
The Octet Rule
The octet rule states that atoms of main-group elements tend to gain, lose, or share enough electrons to form an octet (eight valence electrons). Such noble-gas electron configurations with completely filled valence shells are more stable, and therefore should correspond to how the electrons are arranged in a molecule.
The Lewis diagram for an atom can be used to predict the number of bonds the atom will form. For example, a carbon atom has four valence electrons and therefore requires four more electrons to reach an octet:
It is important to keep in mind that it is impossible to exceed an octet for atoms in the second period. This is particularly relevant because you will encounter numerous molecules containing the elements C, N and O.
Finally, because a hydrogen atom needs only two electrons to fill its valence shell, H is an important exception to the octet rule and forms only one bond.
Exercise: Number of Bonds
Double and Triple Bonds
Two atoms may need to share more than one pair of electrons to achieve the requisite octet. In other words, the bond order is greater than 1. A double bond consists of two pairs of electrons being shared between two atoms. For example:

A triple bond forms when three pairs of electron are shared between two atoms. For example:

Activity: Double and Triple Bonds
Write answers to these questions in your course notebook:
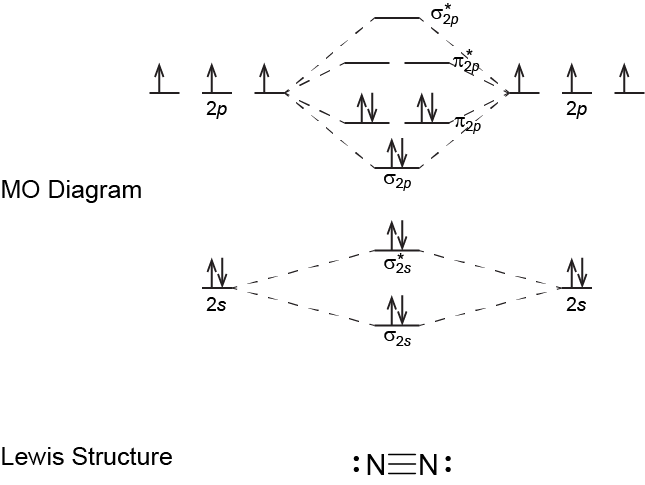
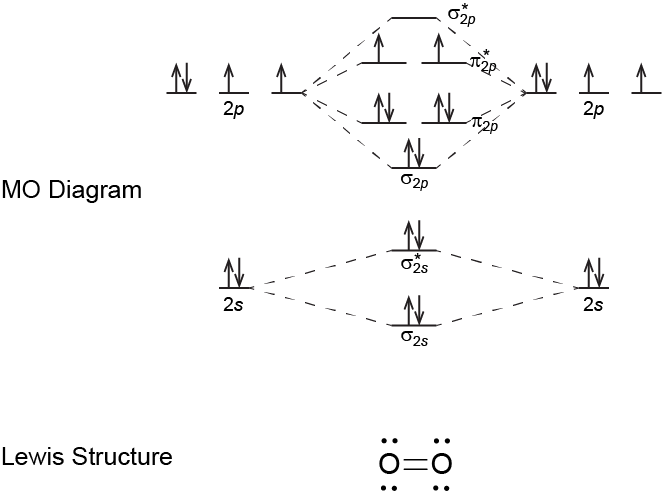
- Write a Lewis structure for N2 and a Lewis structure for O2. Describe the type of bond in each case.
- Do the N atoms in N2 and the O atoms in O2 follow the rule for number of bonds from the above Exercise: Number of Bonds?
- Use the molecular-orbital energy level diagram to calculate the bond order for N2 and for O2. How do the bond orders relate to the Lewis structures?
Write in your notebook, then left-click here for an explanation.
Each N atom forms three bonds (the three electron pairs in the triple bond) and follows the rule for number of bonds. The Lewis structure for N2 also has one lone pair on each N atom.
Each O atom forms two bonds (the two electron pairs in the double bond) and follows the rule for number of bonds. The Lewis structure for O2 also has two lone pairs on each O atom.
The MO diagram for N2 yields a bond order of 3. This corresponds with a triple bond in the Lewis structure.
The MO diagram for O2 gives a bond order of 2. This corresponds with a double bond in the Lewis structure. (Note that the Lewis structure does not predict two unpaired electrons, but the MO diagram does.)
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂 (Note that we cannot answer questions via the google form. If you have a question, please post it on Piazza.)

