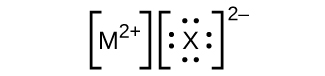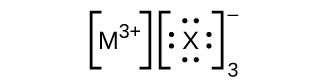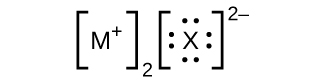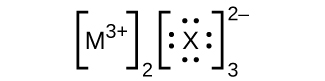45 Lewis Structures (M8Q2)
Introduction
Learning Objectives for Lewis Structures
- Employ the octet rule to draw Lewis structures.
| Lewis Symbols | Lewis Structures | Octet Rule | Writing Lewis Structures with the Octet Rule | - Recognize common exceptions to the octet rule (including radicals) and appraise their impacts on structure and reactivity.
| Exceptions to the Octet Rule |
| Key Concepts and Summary | Glossary | End of Section Exercises |
Lewis Symbols
We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons:
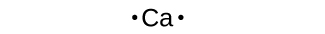
Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table.
![A table is shown that has three columns and nine rows. The header row reads “Atoms,” “Electronic Configuration,” and “Lewis Symbol.” The first column contains the words “sodium,” “magnesium,” “aluminum,” “silicon,” “phosphorus,” “sulfur,” “chlorine,” and “argon.” The second column contains the symbols and numbers “[ N e ] 3 s superscript 2,” “[ N e ] 3 s superscript 2, 3 p superscript 1,” “[ N e ] 3 s superscript 2, 3 p superscript 2,” “[ N e ] 3 s superscript 2, 3 p superscript 3,” “[ N e ] 3 s superscript 2, 3 p superscript 4,” “[ N e ] 3 s superscript 2, 3 p superscript 5,” and “[ N e ] 3 s superscript 2, 3 p superscript 6.” The third column contains Lewis structures for N a with one dot, M g with two dots, A l with three dots, Si with four dots, P with five dots, S with six dots, C l with seven dots, and A r with eight dots.](https://wisc.pb.unizin.org/app/uploads/sites/126/2017/08/CNX_Chem_07_03_3rowLewis.jpg)
Lewis symbols can also be used to illustrate the formation of cations from atoms, as shown here for sodium and calcium:

Likewise, they can be used to show the formation of anions from atoms, as shown here for chlorine and sulfur:

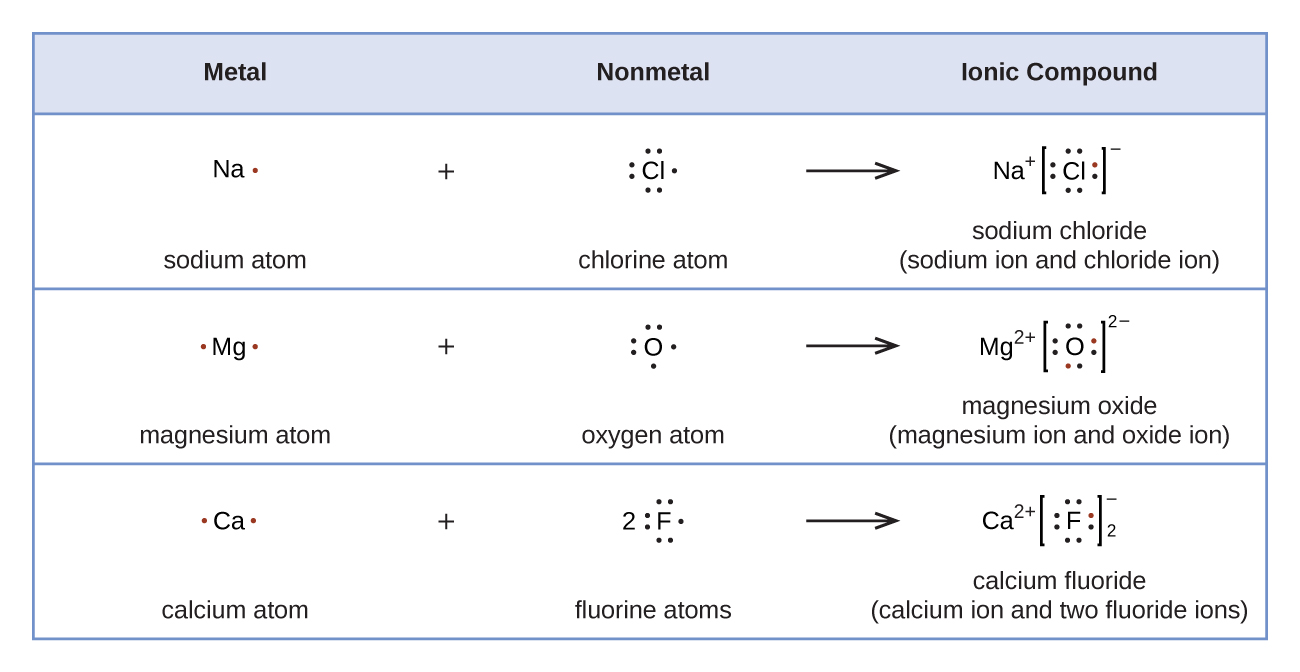
Figure 2 demonstrates the use of Lewis symbols to show the transfer of electrons during the formation of ionic compounds.
Lewis Structures
We also use Lewis symbols to indicate the formation of covalent bonds, which are shown in Lewis structures, drawings that describe the bonding in molecules and polyatomic ions. For example, when two chlorine atoms form a chlorine molecule, they share one pair of electrons:
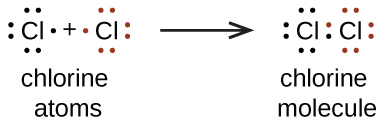
The Lewis structure indicates that each Cl atom has three pairs of electrons that are not used in bonding (called lone pairs) and one shared pair of electrons (written between the atoms). A dash (or line) is the preferred method used to indicate a shared pair of electrons in a covalent bond:

A single shared pair of electrons is called a single bond. Each Cl atom interacts with eight valence electrons: the six in the lone pairs and the two in the single bond.
The Octet Rule
The other halogen molecules (F2, Br2, and I2) form bonds like those in the chlorine molecule: one single bond between atoms and three lone pairs of electrons per atom. This allows each halogen atom to have a noble gas electron configuration. The tendency of main group atoms to form enough bonds to obtain eight valence electrons is known as the octet rule.
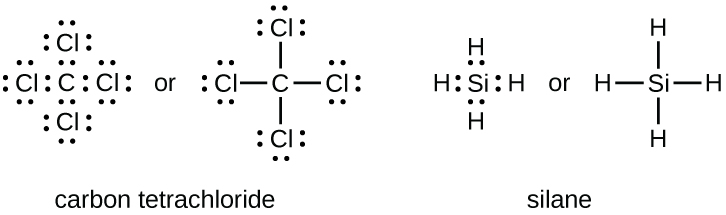
The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons); this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet. These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl4 (carbon tetrachloride) and silicon in SiH4 (silane). Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule. The transition elements and inner transition elements also do not follow the octet rule:
Group 15 elements such as nitrogen have five valence electrons in the atomic Lewis symbol: one lone pair and three unpaired electrons. To obtain an octet, these atoms form three covalent bonds, as in NH3 (ammonia). Oxygen and other atoms in group 16 obtain an octet by forming two covalent bonds. Fluorine and other halogens obtain an octet by forming on covalent bond.
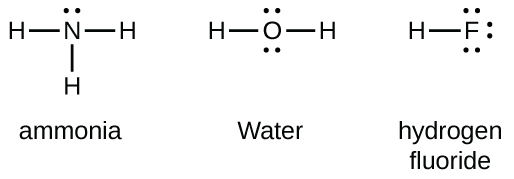
Double and Triple Bonds
As previously mentioned, when a pair of atoms shares one pair of electrons, we call this a single bond. However, a pair of atoms may need to share more than one pair of electrons in order to achieve the requisite octet. A double bond forms when two pairs of electrons are shared between a pair of atoms, as between the carbon and oxygen atoms in CH2O (formaldehyde) and between the two carbon atoms in C2H4 (ethylene):
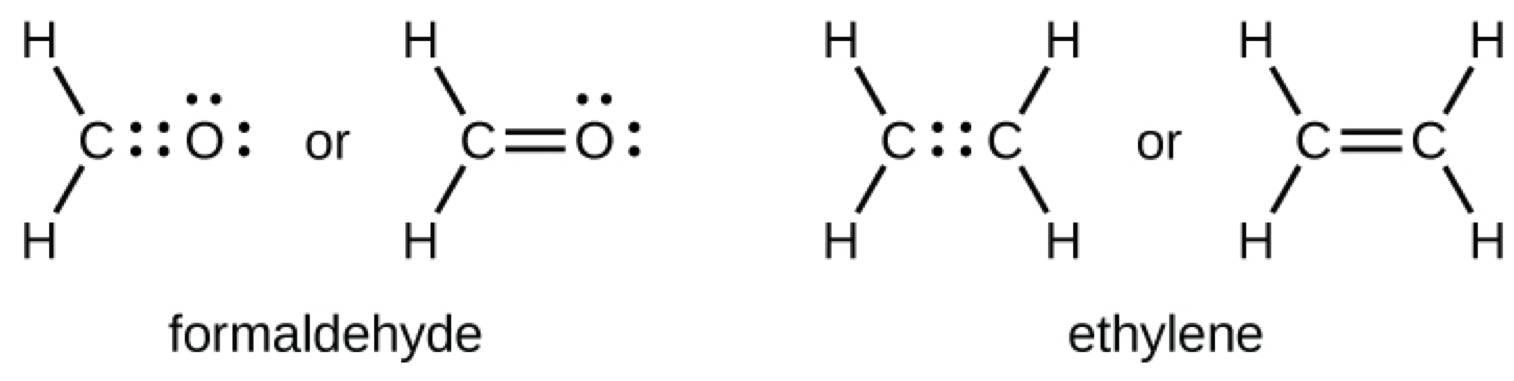
A triple bond forms when three electron pairs are shared by a pair of atoms, as in carbon monoxide (CO) and the cyanide ion (CN–):
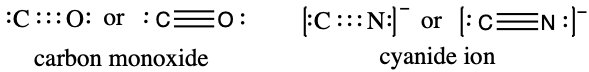
Writing Lewis Structures with the Octet Rule
For very simple molecules and molecular ions, we can write the Lewis structures by merely pairing up the unpaired electrons on the constituent atoms. See these examples:
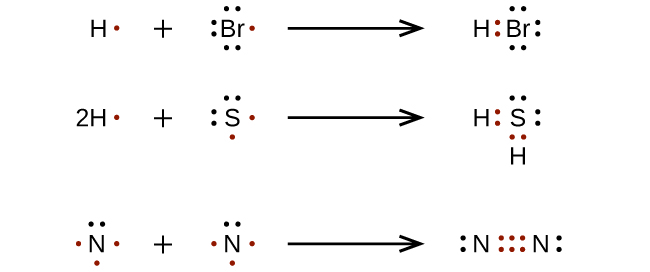
For more complicated molecules and polyatomic ions, it is helpful to follow the step-by-step procedure outlined here:
- Determine the total number of valence (outer shell) electrons for the entire molecule. Recall that the number of valence electrons for an atom comes from what column that atom resides in on the periodic table. Once you have determined the electrons from each atom, you can add them up to determine the total number of valence electrons for the entire molecule.
- If it’s a polyatomic ion, then add or subtract electrons. If it is positively charged you should remove a number of electrons equivalent to the charge. If the polyatomic ion is negatively charged you should add a number of electrons equivalent to the charge.
- Determine the central atom of a molecule.
-
- The central atom of a molecule is often the atom that can form the most bonds.
- It’s also often the least electronegative atom present in the molecule.
- Hydrogen is never the central atom as it can only bond once.
- The central atom is often listed first in the formula.
-
- Attach the remaining atoms by drawing single bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet. Each single bond represents two shared electrons.
- Place the remaining electrons on the central atom(s). If the central atom doesn’t have an octet, then form multiple bonds by moving two unshared electrons from a terminal atom to be shared with the central atom.
- If the molecule is a polyatomic ion, then draw brackets around the structure and write the charge of the molecule outside the brackets. Neutral molecules do not need brackets.
Let us determine the Lewis structures of SiH4, CO32-, NO+, and OF2 as examples, starting with SiH4:
- For SiH4, determine the total number of valence (outer shell) electrons in the molecule or ion.
- For a molecule, we add the number of valence electrons on each atom in the molecule:
+ Si: 4 valence electrons/atom × 1 atom = 4
+ H: 1 valence electron/atom × 4 atoms = 4
= = 8 valence electrons
- For a molecule, we add the number of valence electrons on each atom in the molecule:
- Determine the central atom of the molecule.
- Remember that the central atom of a molecule is often the least electronegative atom, which in this case is silicon, Si.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
- Remember that when you are attaching a terminal atom to the central atom you have to draw a single line between the two atoms. Additionally hydrogen only needs two electrons to be stable so the single bond between silicon and hydrogen is sufficient.

- Place leftover electrons on the central atoms. If the central atom doesn’t have an octet, then create multiple bonds between the central atom and terminal atoms.
- Silicon has an octet already, two electrons from each bond. The correct Lewis structure for this step is the same as the structure depicted above.
- If the molecule is a polyatomic ion, draw a bracket around the molecule and write the charge outside the bracket.
- SiH4 is not ionic, so the final Lewis structure is the same as depicted above.
For CO32- the process is largely the same:
- Determine the total number of valence (outer shell) electrons in the molecule or ion.
- For a negative ion, such as CO32-, we add the number of valence electrons on the atoms, plus two more electrons:
[latex]\begin{array}{r r l} {\text{CO}_3}^{2-} & & \\[1em] & \text{C: 4 valence electrons/atom} \times 1 \;\text{atom} & = 4 \\[1em] & \text{O: 6 valence electrons/atom} \times 3 \;\text{atoms} & = 18 \\[1em] \rule[-0.5ex]{21.5em}{0.1ex}\hspace{-21.5em} + & 2\;\text{additional electrons} & = 2 \\[1em] & & = 24 \;\text{valence electrons} \end{array}[/latex]
- For a negative ion, such as CO32-, we add the number of valence electrons on the atoms, plus two more electrons:
- Determine the central atom of the molecule.
- Carbon is the least electronegative atom in this molecule.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
- Remember that when you are attaching a terminal atom to the central atom you have to draw a single line between the two atoms.
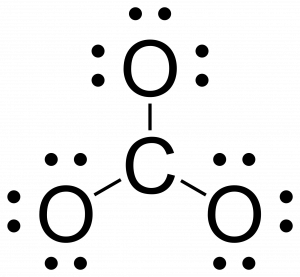
- Place leftover electrons on the central atoms. If the central atom doesn’t have an octet, then create multiple bonds between the central atom and terminal atoms.
- There are no more electrons to place on the central carbon, but carbon does not yet have an octet. To give carbon an octet, we will bind it one more time to one of the currently bonded oxygen atoms. Which atom we choose does not matter in this example, but this will become important for the later topics of resonance structures and pi conjugation.
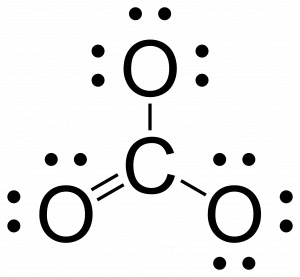
- If the molecule is a polyatomic ion, draw a bracket around the molecule and write the charge outside the bracket.
- CO32- is ionic so we draw brackets around the molecule and denote the charge of 2- outside the brackets.
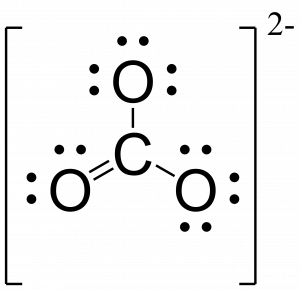
For NO+ the process is as follows:
- Determine the total number of valence (outer shell) electrons in the molecule or ion.
- For a positive ion, such as NO+, we will subtract one electron from the total number of valence electrons:
[latex]\begin{array}{r r l} \text{NO}^{+} & & \\[1em] & \text{N: 5 valence electrons/atom} \times 1 \;\text{atom} & = 5 \\[1em] & \text{O: 6 valence electrons/atom} \times 1 \;\text{atom} & = 6 \\[1em] \rule[-0.5ex]{21em}{0.1ex}\hspace{-21em} + & -1 \;\text{electron (positive charge)} & = -1 \\[1em] & & = 10 \;\text{valence electrons} \end{array}[/latex]
- For a positive ion, such as NO+, we will subtract one electron from the total number of valence electrons:
- Determine the central atom of the molecule.
- Since there are only two atoms in this compound, we don’t refer to one atom as being central.
- Attach the atoms by drawing bonds between the two atoms, then distribute the remaining electron pairs so that one atom has an octet.
- Place leftover electrons on the other atom. If both atoms don’t have an octet, then create multiple bonds between the central atom and terminal atoms.
-
- Nitrogen does not have an octet at this point, so nitrogen needs to bond two more times to oxygen to form a triple bond.
![]()
- If the molecule is a polyatomic ion, draw a bracket around the molecule and write the charge outside the bracket.
- NO+ is ionic so we draw brackets around the molecule and denote the charge of the ion, 1+, outside the brackets.

For OF2 the process is as follows:
- Determine the total number of valence (outer shell) electrons in the molecule or ion.
- Since OF2 is a neutral molecule, we simply add the number of valence electrons:
[latex]\begin{array}{r r l} \text{OF}_{2} & & \\[1em] & \text{O: 6 valence electrons/atom} \times 1 \;\text{atom} & = 6 \\[1em] \rule[-0.5ex]{21em}{0.1ex}\hspace{-21em} + & \text{F: 7 valence electrons/atom} \times 2 \;\text{atoms} & = 14 \\[1em] & & = 20 \;\text{valence electrons} \end{array}[/latex]
- Since OF2 is a neutral molecule, we simply add the number of valence electrons:
- Determine the central atom of the molecule.
- Oxygen is less electronegative than fluorine, so oxygen is the central atom.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
- Remember that when you are attaching a terminal atom to the central atom you have to draw a single line between the two atoms.
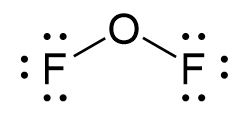
- Place leftover electrons on the other atom. If both atoms don’t have an octet, then create multiple bonds between the central atom and terminal atoms.
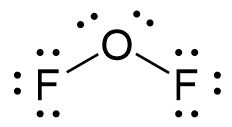
- If the molecule is a polyatomic ion, draw a bracket around the molecule and write the charge outside the bracket.
- OF2 is not ionic so the final Lewis structure is the same as depicted above.
Example 1
Writing Lewis Structures
NASA’s Cassini-Huygens mission detected a large cloud of toxic hydrogen cyanide (HCN) on Titan, one of Saturn’s moons. Titan also contains ethane (H3CCH3), acetylene (HCCH), and ammonia (NH3). What are the Lewis structures of these molecules?
Solution
HCN
- Calculate the number of valence electrons. (1 × 1) + (4 × 1) + (5 × 1) = 10
- Determine the central atom of the molecule: Carbon is the least electronegative, so carbon is the central atom.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
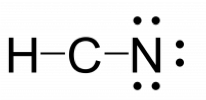
- Place the remaining electron pairs on the central atom. If the central atom doesn’t have an octet create multiple bonds between the central and terminal atoms.
- HCN does not have a complete octet around the carbon so two additional bonds are needed.
![]()
- If the molecule was ionic, draw brackets around the Lewis structure and write the charge outside the bracket. HCN is not ionic, so no brackets are necessary and the above structure is the final Lewis structure.
H3CCH3
- Calculate the number of valence electrons. (1 × 3) + (2 × 4) + (1 × 3) = 14
- Determine the central atom of the molecule: Carbon is the least electronegative, so the carbon atoms are the central atoms.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
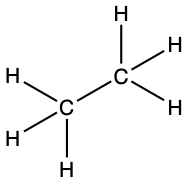
-
- We can consider the multiple carbon atoms in H3CCH3 as all being the central atoms since they are equivalent.
- Place the remaining electron pairs on the central atom. If the central atom doesn’t have an octet create multiple bonds between the central and terminal atoms.
- If the molecule was ionic, draw brackets around the Lewis structure and write the charge outside the bracket. H3CCH3 is not ionic, so no brackets are necessary and the above structure is the final Lewis structure.
HCCH
- Calculate the number of valence electrons. (1 × 1) + (2 × 4) + (1 × 1) = 10
- Determine the central atom of the molecule: Carbon is the least electronegative, so the carbon atoms are the central atoms.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
![]()
-
- We can consider the multiple carbon atoms in HCCH as all being the central atoms since they are equivalent.
- Place the remaining electron pairs on the central atom. If the central atom doesn’t have an octet create multiple bonds between the central and terminal atoms. The central atoms have octets.
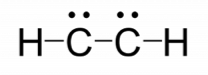
-
- HCCH does not have a complete octet around each carbon so additional bonds are needed.
![]()
- If the molecule was ionic, draw brackets around the Lewis structure and write the charge outside the bracket. HCCH is not ionic, so no brackets are necessary and the above structure is the final Lewis structure.
NH3
- Calculate the number of valence electrons. (5 × 1) + (3 × 1) = 8
- Determine the central atom of the molecule: Nitrogen is the least electronegative, so the nitrogen atom is the central atom.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
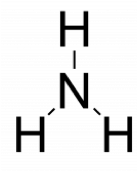
- Place the remaining electron pairs on the central atom. If the central atom doesn’t have an octet create multiple bonds between the central and terminal atoms. The central atoms have octets.
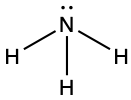
- If the molecule was ionic, draw brackets around the Lewis structure and write the charge outside the bracket. NH3 is not ionic, so no brackets are necessary and the above structure is the final Lewis structure.
Check Your Learning
Both carbon monoxide, CO, and carbon dioxide, CO2, are products of the combustion of fossil fuels. Both of these gases also cause problems: CO is toxic and CO2 has been implicated in global climate change. What are the Lewis structures of these two molecules?
Answer:
![]()
Exceptions to the Octet Rule
Many covalent molecules have central atoms that do not have eight electrons in their Lewis structures. These molecules fall into three categories:
- Electron-deficient molecules have a central atom that has fewer than eight electrons.
- Hypervalent molecules have a central atom that has more than eight electrons.
- Free radicals have an odd number of valence electrons, and therefore have an unpaired electron.
Electron-deficient Molecules
We will encounter a few molecules that contain central atoms that do not have an octet of valence electrons, such as hydrogen, beryllium, and boron in BH3. Generally, these are molecules that contain central atoms from groups 2 and 13, outer atoms that are hydrogen, or other atoms that do not form multiple bonds. For example, in the Lewis structures of beryllium dihydride, BeH2, and boron trihydride, BH3, the beryllium and boron atoms each have only four and six electrons, respectively.
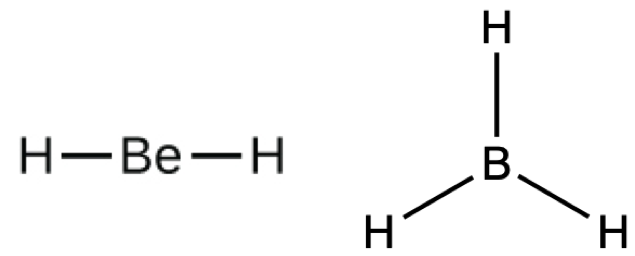
Hypervalent Molecules
Elements in the second period of the periodic table (n = 2) can accommodate a maximum of eight electrons in their valence shell orbitals because they have only four valence orbitals (one 2s and three 2p orbitals). Elements in the third and higher periods (n ≥ 3) can accommodate more than four pairs of electrons in their valence shells as lone pairs or bonded pairs shared with other atoms. Molecules formed from these elements are sometimes called hypervalent molecules. Figure 3 shows the Lewis structures for two hypervalent molecules, PCl5 and SF6.
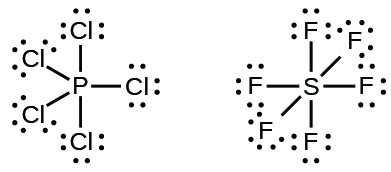
In some hypervalent molecules, such as IF5 and XeF4, some of the electrons in the outer shell of the central atom are lone pairs:
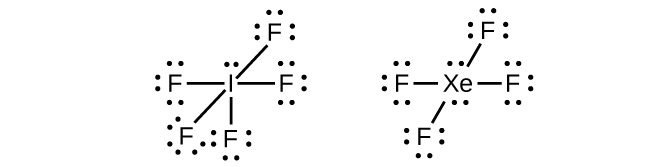
When we write the Lewis structures for these molecules, we find that we have electrons left over after filling the valence shells of the outer atoms with eight electrons. These additional electrons must be assigned to the central atom.
Example 2
Writing Lewis Structures: Octet Rule Violations
Xenon is a noble gas, but it forms a number of stable compounds. We examined XeF4 earlier. What are the Lewis structures of XeF2 and XeF6?
Solution
We can draw the Lewis structure of any covalent molecule by following the five steps discussed earlier. In this case, we can condense the last few steps, since not all of them apply.
XeF2
- Calculate the number of valence electrons: 8 + (2 × 7) = 22
- Determine the central atom of the molecule: The central atom is xenon since noble gases do not want any additional electrons, as they have fully filled subshells, and thus have very low electronegativities.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
![]()
- Place the remaining electron pairs on the central atom. If the central atom doesn’t have an octet create multiple bonds between the central and terminal atoms. In this instance the xenon atom will be hypervalent and have more than eight electrons surrounding it, violating the octet rule.
![]()
- If the molecule was ionic, draw brackets around the Lewis structure and write the charge outside the bracket.
- This molecule is not ionic so the final Lewis structure is the same as depicted above.
XeF6
- Calculate the number of valence electrons: XeF6: 8 + (6 × 7) = 50
- Determine the central atom of the molecule: The central atom is xenon since noble gases do not want any additional electrons, as they have fully filled subshells, and thus have very low electronegativities.
- Attach the remaining atoms by drawing bonds between the central atom and terminal atoms, then distribute the remaining electron pairs so that all terminal atoms have an octet.
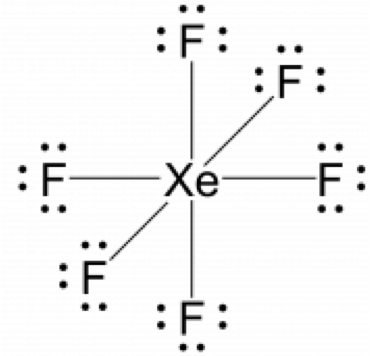
- Place the remaining electron pairs on the central atom. If the central atom doesn’t have an octet create multiple bonds between the central and terminal atoms. In this instance the xenon atoms will be hypervalent and have more than eight electrons surrounding it, violating the octet rule.
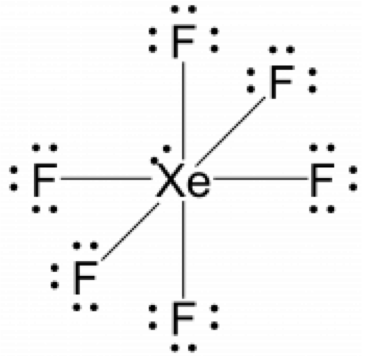
- If the molecule was ionic, draw brackets around the Lewis structure and write the charge outside the bracket.
- This molecule is not ionic so the final Lewis structure is the same as depicted above.
Check Your Learning
The halogens form a class of compounds called the interhalogens, in which halogen atoms covalently bond to each other. Write the Lewis structures for the interhalogens BrCl3 and ICl4−.
Answer:
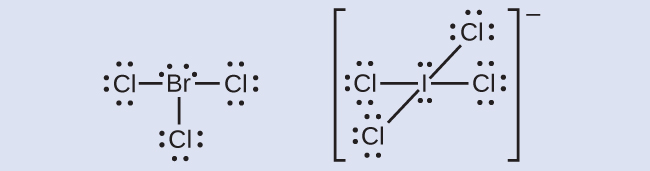
Odd-electron Molecules
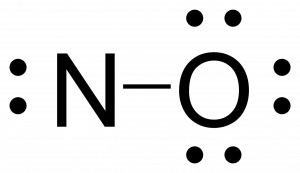
Free radicals are neutral molecules that contain an unpaired electron often as a result of possessing an odd number of valence electrons. Nitric oxide, NO, is an example of an odd-electron molecule; it is produced in internal combustion engines when oxygen and nitrogen react at high temperatures.
To draw the Lewis structure for an odd-electron molecule like NO, we follow the same five steps we would for other molecules, but the results of these steps might look slightly different.
- Calculate the number of valence electrons. The sum of the valence electrons is 5 (from N) + 6 (from O) = 11. The odd number immediately tells us that we have a free radical, so we know that not every atom can have an octet in the Lewis structure.
- Determine the central atom of the molecule: Since there are only two atoms in this compound, we don’t refer to one atom as being central.
- Attach the remaining atoms by drawing bonds between the atoms, then distribute the remaining electron pairs so that all atoms have an octet.
- If all atoms don’t have an octet create multiple bonds between the central and terminal atoms. Since we have an odd number of total electrons, an atom must carry the free radical electron. While all atoms can have the unpaired electron, it is generally better to place it on the least electronegative atom. This will be explored in the module on resonance structures.
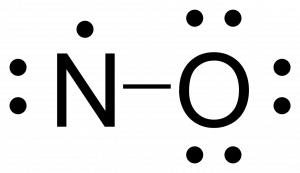
-
- Nitrogen only has five electrons around it, so nitrogen needs another bond to have seven valence electrons. Since the total number of valence electrons is 11, nitrogen will never have an octet. This is important as molecules with free radicals are highly reactive due to their propensity to form a closed shell.

- If the molecule was ionic, draw brackets around the Lewis structure and write the charge outside the bracket.
- NO is not ionic so the final Lewis structure is the same as depicted above.
Key Concepts and Summary
Valence electronic structures can be visualized by drawing Lewis symbols (for atoms and monatomic ions) and Lewis structures (for molecules and polyatomic ions). Lone pairs, unpaired electrons, and single, double, or triple bonds are used to indicate where the valence electrons are located around each atom in a Lewis structure. Most structures—especially those containing second row elements—obey the octet rule, in which every atom (except H) is surrounded by eight electrons. Exceptions to the octet rule occur for odd-electron molecules (free radicals), electron-deficient molecules, and hypervalent molecules.
Glossary
- double bond
- covalent bond in which two pairs of electrons are shared between two atoms
- free radical
- molecules that contains an unpaired electron
- hypervalent molecule
- molecule containing at least one main group element that has more than eight electrons in its valence shell
- Lewis structure
- diagram showing lone pairs and bonding pairs of electrons in a molecule or an ion
- Lewis symbol
- symbol for an element or monatomic ion that uses a dot to represent each valence electron in the element or ion
- lone pair
- two (a pair of) valence electrons that are not used to form a covalent bond
- octet rule
- guideline that states main group atoms will form structures in which eight valence electrons interact with each nucleus, counting bonding electrons as interacting with both atoms connected by the bond
- single bond
- bond in which a single pair of electrons is shared between two atoms
- triple bond
- bond in which three pairs of electrons are shared between two atoms
Chemistry End of Section Exercises
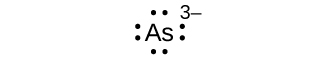
- Write the Lewis symbols for each of the following ions:
- As3–
- I–
- Be2+
- O2–
- Ga3+
- Li+
- N3–
- Write the Lewis symbols of the ions in each of the following ionic compounds:
- MgS
- Al2O3
- GaCl3
- K2O
- Li3N
- KF
- In the Lewis structures listed here, M and X represent various elements in the third period of the periodic table. Write the formula of each compound using the chemical symbols of each element:
- Write the Lewis structure for the diatomic molecule P2, an unstable form of phosphorus found in high-temperature phosphorus vapor.
- Write Lewis structures for the following:
- O2
- H2CO
- AsF3
- ClNO
- SiCl4
- H3O+
- NH4+
- BF4−
- HCCH
- ClCN
- C22+
- Write Lewis structures for the following:
- SeF6
- XeF4
- SeCl3+
- Cl2BBCl2 (contains a B–B bond)
- Write Lewis structures for:
- PO43−
- ICl4−
- SO32−
- HONO
- Correct the following statement: “The bonds in solid PbCl2 are ionic; the bond in a HCl molecule is covalent. Thus, all of the valence electrons in PbCl2 are located on the Cl– ions, and all of the valence electrons in a HCl molecule are shared between the H and Cl atoms.”
- Methanol, H3COH, is used as the fuel in some race cars. Ethanol, C2H5OH, is used extensively as motor fuel in Brazil. Both methanol and ethanol produce CO2 and H2O when they burn. Write the chemical equations for these combustion reactions using Lewis structures instead of chemical formulas.
- Many planets in our solar system contain organic chemicals including methane (CH4) and traces of ethylene (C2H4), ethane (C2H6), propyne (H3CCCH), and diacetylene (HCCCCH). Write the Lewis structures for each of these molecules.
- Carbon tetrachloride was formerly used in fire extinguishers for electrical fires. It is no longer used for this purpose because of the formation of the toxic gas phosgene, Cl2CO. Write the Lewis structures for carbon tetrachloride and phosgene.
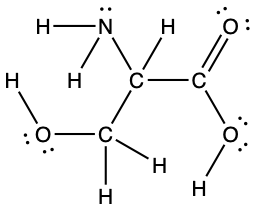
- The arrangement of atoms in several biologically important molecules is given here. Complete the Lewis structures of these molecules by adding multiple bonds and lone pairs. Do not add any more atoms.
- the amino acid serine:
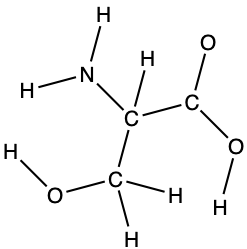
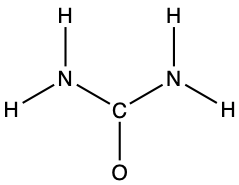
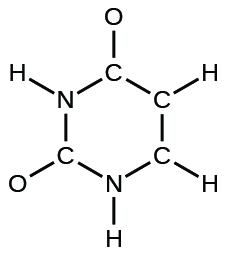
- urea:
- pyruvic acid:
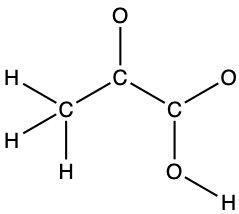
- uracil:
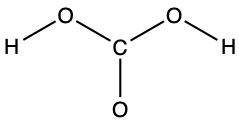
- carbonic acid:
- the amino acid serine:
- A compound with a molar mass of about 42 g/mol contains 85.7% carbon and 14.3% hydrogen by mass. Write the Lewis structure for a molecule of the compound.
- Two arrangements of atoms are possible for a compound with a molar mass of about 45 g/mol that contains 52.2% C, 13.1% H, and 34.7% O by mass. Write the Lewis structures for the two molecules.
- How are single, double, and triple bonds similar? How do they differ?
Answers to Chemistry End of Section Exercises
- (a) eight electrons:
 ;
;
(b) eight electrons: ;
;
(c) no electrons: Be2+;
(d) eight electrons: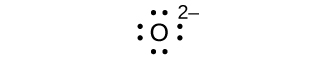 ;
;
(e) no electrons: Ga3+;
(f) no electrons: Li+;
(g) eight electrons: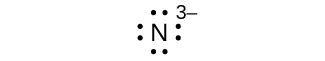
- (a)
 ;
;
(b) ;
;
(c)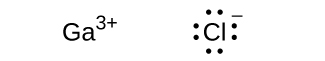 ;
;
(d)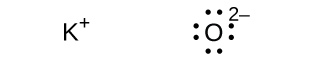 ;
;
(e)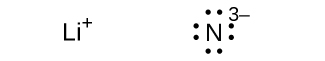 ;
;
(f)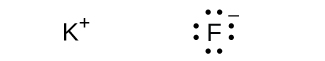
- (a) MgS; (b) AlCl3; (c) Na2S; (d) Al2S3

- (a)
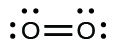 (In this case, the Lewis structure is inadequate to depict the fact that experimental studies have shown two unpaired electrons in each oxygen molecule);
(In this case, the Lewis structure is inadequate to depict the fact that experimental studies have shown two unpaired electrons in each oxygen molecule);
(b)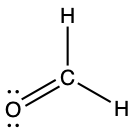 ;
;
(c)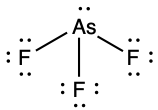 ;
;
(d)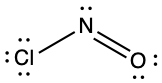 ;
;
(e)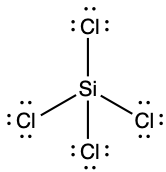 ;
;
(f)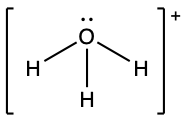 ;
;
(g)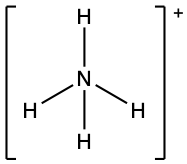 ;
;
(h)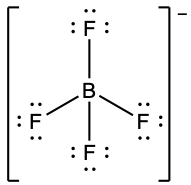 ;
;
(i)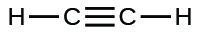 ;
;
(j)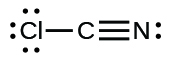 ;
;
(k)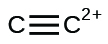
- (a)
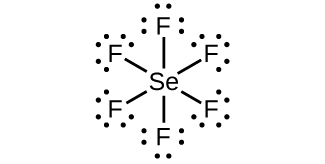 ;
;
(b)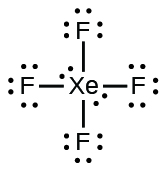 ;
;
(c)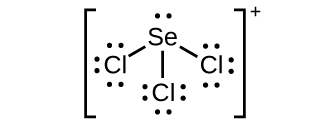 ;
;
(d)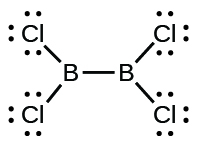
- (a)
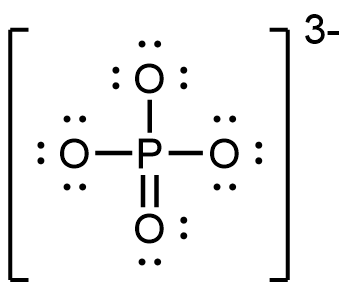 ;
;
(b)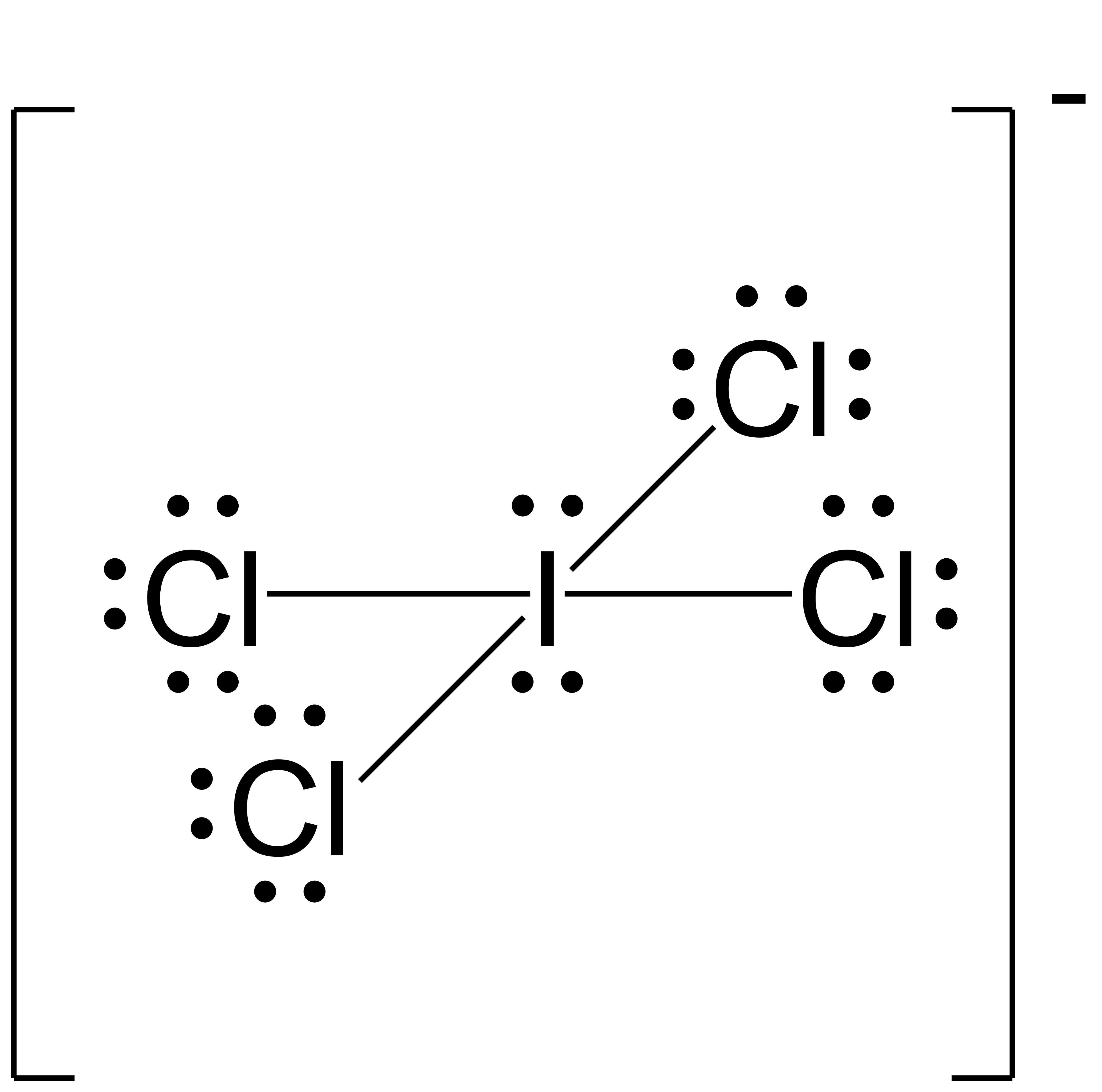 ;
;
(c)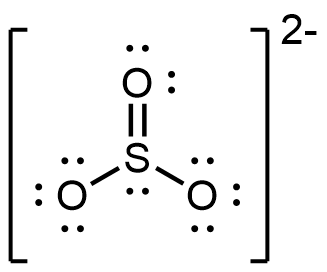 ;
;
(d)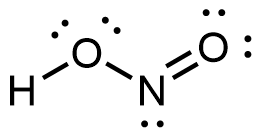
- Two valence electrons per Pb atom are transferred to Cl atoms; the resulting Pb2+ ion has a 6s2 valence shell configuration. Two of the valence electrons in the HCl molecule are shared, and the other six are located on the Cl atom as lone pairs of electrons.
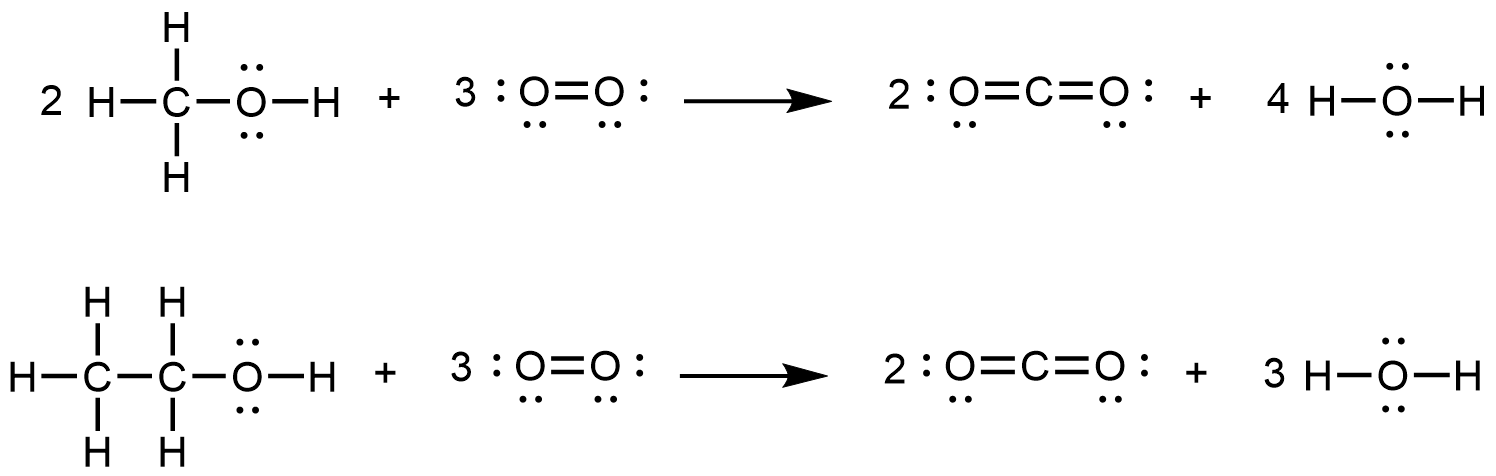
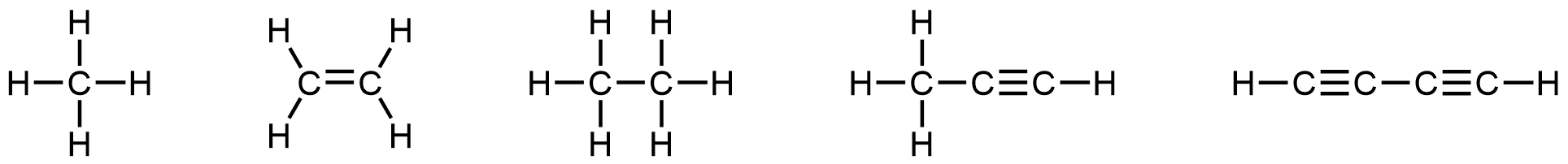
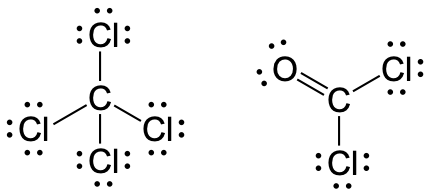
- (a)
 ;
;
(b)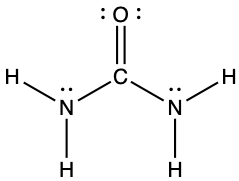 ;
;
(c)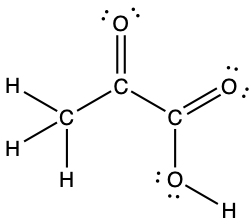 ;
;
(d)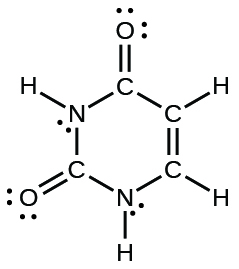 ;
;
(e)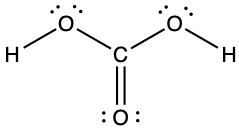
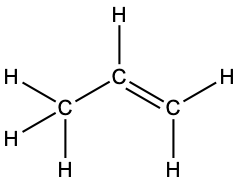

- Each bond includes a sharing of electrons between atoms. Two electrons are shared in a single bond; four electrons are shared in a double bond; and six electrons are shared in a triple bond.