M15Q4: Autoionization and the pH Scale
Learning Objectives
- Recognize that water can autoionize and describe the relationship between [H+], [OH–] and Kw.
- Apply the relationship between the pH scale and [H+] and the relationship between pH and pOH as appropriate.
| pH Scale | - Calculate the pH of strong and weak acid and base solutions.
| pH Calculations |
| Key Concepts and Summary | Key Equations | Glossary | End of Section Exercises |
In the preceding sections, we saw that water can function as either an acid or a base, depending on the nature of the solute dissolved in it. In fact, in pure water or in any aqueous solution, water acts both as an acid and a base. A very small fraction of water molecules donate protons to other water molecules to form hydronium ions and hydroxide ions:
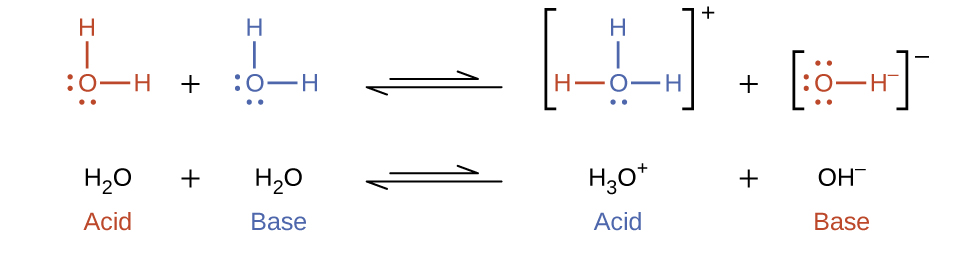
This type of reaction, in which a substance ionizes when one molecule of the substance reacts with another molecule of the same substance, is referred to as autoionization.
Autoionization of Water
Consider the ionization reactions for a conjugate acid-base pair, HA − A−:
HA(aq) + H2O(ℓ) ⇌ H3O+(aq) + A–(aq) Ka = ![]()
A–(aq) + H2O(ℓ) ⇌ OH–(aq) + HA(aq) Kb = ![]()
Adding these two chemical equations yields the equation for the autoionization for water:
HA(aq) + H2O(ℓ) + A–(aq) + H2O(ℓ) ⇌ H3O+(aq) + A–(aq) + OH–(aq) + HA(aq)
2 H2O(ℓ) ⇌ H3O+(aq) + OH–(aq)
As shown in the previous chapter on equilibrium, the K expression for a chemical equation derived from adding two or more other chemical equations is the product of the other equations’ K expressions. Multiplying the equilibrium expressions together and cancelling common terms, we see that:
Ka × Kb = ![]() = [H3O+][OH–] = Kw
= [H3O+][OH–] = Kw
For example, the acid ionization constant of acetic acid (CH3COOH) is 1.8 × 10−5, and the base ionization constant of its conjugate base, acetate ion (CH3COO–), is 5.6 × 10−10. The product of these two constants is called the ion-product constant for water (Kw):
Ka × Kb = (1.8 × 10-5) × (5.6 × 10-10) = 1.0 × 10-14 = Kw
The slight ionization of pure water is reflected in the small value of the equilibrium constant; at 25 °C, Kw has a value of 1.0 × 10−14. The process is endothermic, and so the extent of ionization and the resulting concentrations of hydronium ion and hydroxide ion increase with temperature. For example, at 100 °C, the value for Kw is about 5.6 × 10−13, roughly 50 times larger than the value at 25 °C.
Example 1
Ion Concentrations in Pure Water
What are the hydronium ion concentration and the hydroxide ion concentration in pure water at 25 °C?
Solution
The autoionization of water yields the same number of hydronium and hydroxide ions. Therefore, in pure water, [H3O+] = [OH−] = “x“. At 25 °C:
Kw = [H3O+][OH–] = x2 = 1.0 × 10-14
So:
[H3O+] = [OH–] = (1.0 × 10-14)½ = 1.0 × 10-7 M
The hydronium ion concentration and the hydroxide ion concentration are the same, and we find that both equal 1.0 × 10−7 M.
Check Your Learning
The ion product of water at 80 °C is 2.4 × 10−13. What are the concentrations of hydronium and hydroxide ions in pure water at 80 °C?
Answer:
[H3O+] = [OH−] = 4.9 × 10−7 M
It is important to realize that the autoionization equilibrium for water is established in all aqueous solutions. Adding an acid or base to water will not change the position of the equilibrium. Example 2 demonstrates the quantitative aspects of this relation between hydronium and hydroxide ion concentrations.
Example 2
The Inverse Proportionality of [H3O+] and [OH−]
A solution of carbon dioxide in water has a hydronium ion concentration of 2.0 × 10−6 M. What is the concentration of hydroxide ion at 25 °C?
Solution
We know the value of the ion-product constant for water at 25 °C:
2 H2O(ℓ) ⇌ H3O+(aq) + OH–(aq) Kw = [H3O+][OH–] = 1.0 × 10-14
Thus, we can calculate the missing equilibrium concentration.
Rearrangement of the Kw expression yields that [OH−] is directly proportional to the inverse of [H3O+]:
[OH–] = ![]() = 5.0 × 10-9
= 5.0 × 10-9
The hydroxide ion concentration in water is reduced to 5.0 × 10−9 M as the hydrogen ion concentration increases to 2.0 × 10−6M. This is expected from Le Châtelier’s principle; the autoionization reaction shifts to the left to reduce the stress of the increased hydronium ion concentration and the [OH−] is reduced relative to that in pure water.
A check of these concentrations confirms that our arithmetic is correct:
Kw = [H3O+][OH–] = (2.0 × 10-6)(5.0 × 10-9) = 1.0 × 10-14
Check Your Learning
What is the hydronium ion concentration in an aqueous solution with a hydroxide ion concentration of 0.001 M at 25 °C?
Answer:
[H3O+] = 1 × 10−11 M
As discussed earlier, hydronium and hydroxide ions are present both in pure water and in all aqueous solutions, and their concentrations are inversely proportional as determined by the ion product of water (Kw). The concentrations of these ions in a solution are often critical determinants of the solution’s properties and the chemical behaviors of its other solutes, and specific vocabulary has been developed to describe these concentrations in relative terms. A solution is neutral if it contains equal concentrations of hydronium and hydroxide ions; acidic if it contains a greater concentration of hydronium ions than hydroxide ions; and basic if it contains a lesser concentration of hydronium ions than hydroxide ions.
A common means of expressing quantities, the values of which may span many orders of magnitude, is to use a logarithmic scale. One such scale that is very popular for chemical concentrations and equilibrium constants is based on the p-function, defined as shown where “X” is the quantity of interest and “log” is the base-10 logarithm:
pX = -logX
The pH of a solution is therefore defined as shown here, where [H3O+] is the molar concentration of hydronium ion in the solution:
pH = -log[H3O+]
Rearranging this equation to isolate the hydronium ion molarity yields the equivalent expression:
[H3O+] = 10-pH
Likewise, the hydroxide ion molarity may be expressed as a p-function, or pOH:
pOH = -log[OH–]
or
[OH–] = 10-pOH
Finally, the relation between these two ion concentration expressed as p-functions is easily derived from the Kw expression:
Kw = [H3O+][OH–]
-log(Kw) = -log([H3O+][OH–]) = (-log[H3O+]) + (-log[OH–])
pKw = pH + pOH
At 25 °C, the value of Kw is 1.0 × 10−14, and so:
14.00 = pH + pOH
All of these equations are connected in such a way that if you have the [H3O+], [OH–], pH, or pOH, you can solve for the other three:
![This figure is a square with four double-headed arrows pointing between the four corners along the edges. The top left corner is labeled [H 3 O +]. The arrow moving left is labeled "[H 3 O +] [O H -] = 1.0 times 10 ^ -14". The top right corner is labeled [O H -]. The arrow pointing down is labeled "p O H = - log [O H -]." The bottom right corner is labeled p O H. The arrow pointing left is labeled "p H + p O H = 14.00". The bottom left corner is labeled p H. The arrow pointing up is labeled "p H = - log [ H 3 O +]".](https://wisc.pb.unizin.org/app/uploads/sites/557/2020/10/Chem104_M4Q4_pHsquare.png)
As was shown above in Example 1, the hydronium ion molarity in pure water (or any neutral solution) is 1.0 × 10−7M at 25 °C. The pH and pOH of a neutral solution at this temperature are therefore:
pH = -log[H3O+] = -log[1.0 × 10−7] = 7.00
pOH = -log[OH–] = -log[1.0 × 10−7] = 7.00
And so, at this temperature, acidic solutions are those with hydronium ion molarities greater than 1.0 × 10−7 M and hydroxide ion molarities less than 1.0 × 10−7 M (corresponding to pH values less than 7.00 and pOH values greater than 7.00). Basic solutions are those with hydronium ion molarities less than 1.0 × 10−7 M and hydroxide ion molarities greater than 1.0 × 10−7 M (corresponding to pH values greater than 7.00 and pOH values less than 7.00).
Since the autoionization constant Kw is temperature dependent, these correlations between pH values and the acidic/neutral/basic adjectives will be different at temperatures other than 25 °C. For example, the “Check Your Learning” exercise accompanying Example 1 in this section showed the hydronium molarity of pure water at 80 °C is 4.9 × 10−7 M, which corresponds to pH and pOH values of:
pH = -log[H3O+] = -log[4.9 × 10−7] = 6.31
pOH = -log[OH–] = -log[4.9 × 10−7] = 6.31
At this temperature, then, neutral solutions exhibit pH = pOH = 6.31, acidic solutions exhibit pH less than 6.31 and pOH greater than 6.31, whereas basic solutions exhibit pH greater than 6.31 and pOH less than 6.31. This distinction can be important when studying certain processes that occur at nonstandard temperatures, such as enzyme reactions in warm-blooded organisms. Unless otherwise noted, references to pH values are presumed to be those at standard temperature (25 °C) (Table 1).
| Classification | Relative Ion Concentrations | pH at 25 °C |
|---|---|---|
| acidic | [H3O+] > [OH−] | pH < 7 |
| neutral | [H3O+] = [OH−] | pH = 7 |
| basic | [H3O+] < [OH−] | pH > 7 |
Figure 1 shows the relationships between [H3O+], [OH−], pH, and pOH, and gives values for these properties at standard temperatures for some common substances.
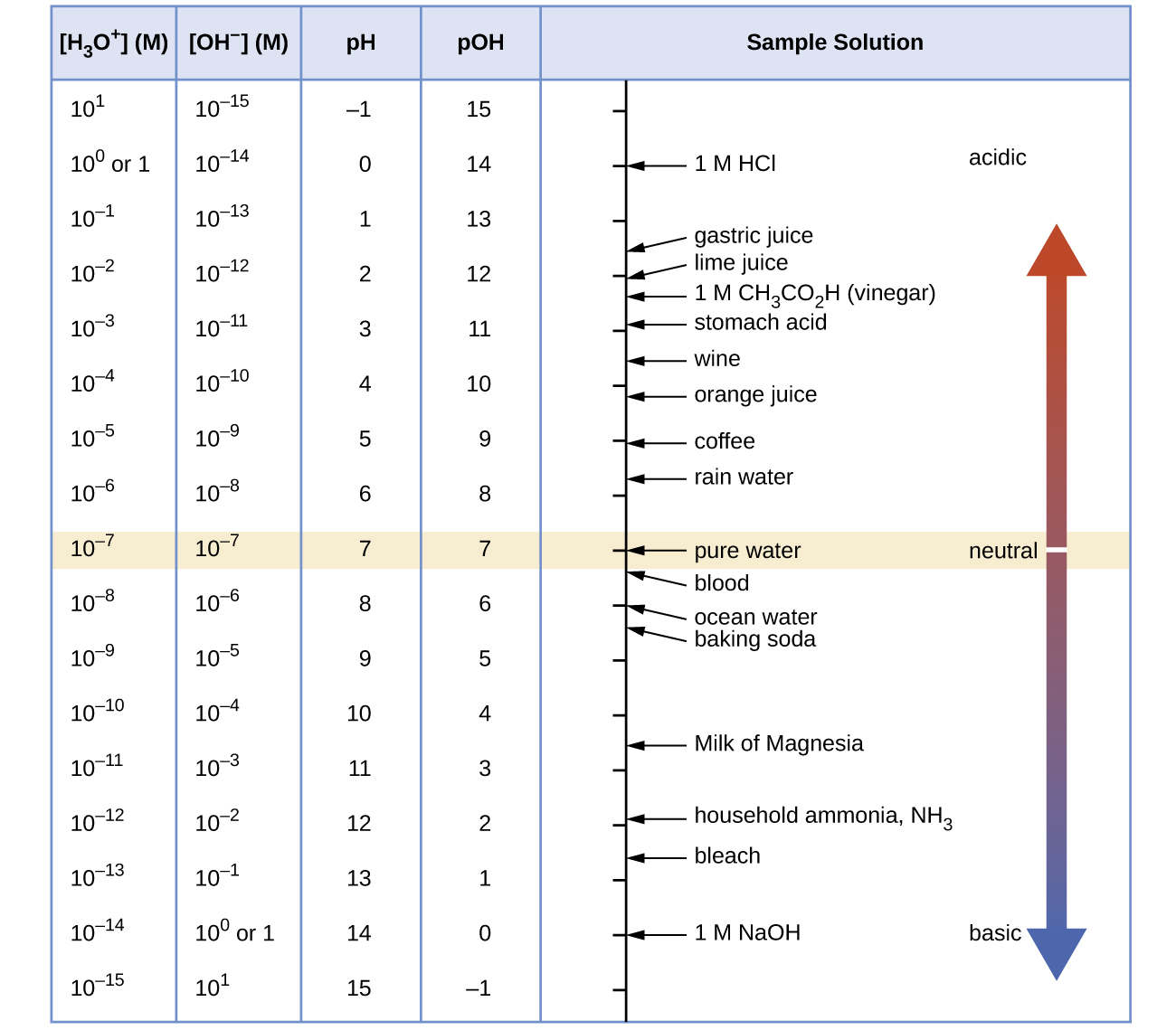
Example 3
Calculation of pH from [H3O+]
What is the pH of stomach acid, a solution of HCl with a hydronium ion concentration of 1.2 × 10−3 M?
Solution
pH = -log[H3O+] = -log(1.2 × 10-3) = -(-2.92) = 2.92
(The use of logarithms is explained in Appendix B. Recall that, as we have done here, when taking the log of a value, keep as many decimal places in the result as there are significant figures in the value.)
Check Your Learning
Water exposed to air contains carbonic acid, H2CO3, due to the reaction between carbon dioxide and water:
CO2(aq) + H2O(ℓ) ⇌ H2CO3(aq)
Air-saturated water has a hydronium ion concentration caused by the dissolved CO2 of 2.0 × 10−6M, about 20-times larger than that of pure water. Calculate the pH of the solution at 25 °C.
Answer:
5.70
Example 4
Calculation of Hydronium Ion Concentration from pH
Calculate the hydronium ion concentration of blood, the pH of which is 7.3 (slightly alkaline).
Solution
pH = -log[H3O+] = 7.3
log[H3O+] = -7.3
[H3O+] = 10-7.3 = 5 × 10-8 M
Check Your Learning
Calculate the hydronium ion concentration of a solution with a pH of −1.07.
Answer:
12 M
Example 5
Calculation of pOH
What are the pOH and the pH of a 0.0125 M solution of potassium hydroxide, KOH?
Solution
Potassium hydroxide is a highly soluble ionic compound and completely dissociates when dissolved in dilute solution, yielding [OH−] = 0.0125 M:
pOH = -log[OH–] = -log(0.0125) = -(-1.903) = 1.903
The pH can be found from the pOH:
pH + pOH = 14.000
pH = 14.000 – pOH = 14.000 – 1.903 = 12.097
Check Your Learning
The hydronium ion concentration of vinegar is approximately 4 × 10−3 M. What are the corresponding values of pOH and pH?
Answer:
pOH = 11.6, pH = 2.4
The acidity of a solution is typically assessed experimentally by measurement of its pH. The pOH of a solution is not usually measured, as it is easily calculated from an experimentally determined pH value. The pH of a solution can be directly measured using a pH meter (Figure 2).

The pH of a solution may also be visually estimated using colored indicators (Figure 3).

Chemistry in Real Life: Environmental Science
Normal rainwater has a pH between 5 and 6 due to the presence of dissolved CO2 which forms carbonic acid:
H2O(ℓ) + CO2(g) → H2CO3(aq)
H2CO3(aq) ⇌ H+(aq) + HCO3–(aq)
Acid rain is rainwater that has a pH of less than 5, due to a variety of nonmetal oxides, including CO2, SO2, SO3, NO, and NO2 being dissolved in the water and reacting with it to form not only carbonic acid, but sulfuric acid and nitric acid. The formation and subsequent ionization of sulfuric acid are shown here:
H2O(ℓ) + SO3(g) → H2SO4(aq)
H2SO4(aq) → H+(aq) + HSO4–(aq)
Carbon dioxide is naturally present in the atmosphere because we and most other organisms produce it as a waste product of metabolism. Carbon dioxide is also formed when fires release carbon stored in vegetation or when we burn wood or fossil fuels. Sulfur trioxide in the atmosphere is naturally produced by volcanic activity, but it also stems from burning fossil fuels, which have traces of sulfur, and from the process of “roasting” ores of metal sulfides in metal-refining processes. Oxides of nitrogen are formed in internal combustion engines where the high temperatures make it possible for the nitrogen and oxygen in air to chemically combine.
Acid rain is a particular problem in industrial areas where the products of combustion and smelting are released into the air without being stripped of sulfur and nitrogen oxides. In North America and Europe until the 1980s, it was responsible for the destruction of forests and freshwater lakes, when the acidity of the rain actually killed trees, damaged soil, and made lakes uninhabitable for all but the most acid-tolerant species. Acid rain also corrodes statuary and building facades that are made of marble and limestone (Figure 4). Regulations limiting the amount of sulfur and nitrogen oxides that can be released into the atmosphere by industry and automobiles have reduced the severity of acid damage to both natural and manmade environments in North America and Europe. It is now a growing problem in industrial areas of China and India.
For further information on acid rain, visit this website hosted by the US Environmental Protection Agency.

Key Concepts and Summary
Although we can conceptually determine which of two molecules is a stronger acid or base, quantitative methods are even more robust. In this section, we introduce the concept of p_, where p stands for the -log of a quantity. The pH is the -log[H3O+], where the [H3O+] is the concentration of hydronium at equilibrium. The same is true for pOH, where pOH = -log[OH–]. If one of these four variables are known (pH, pOH, [H3O+], or [OH–]), the other three can be calculated using the equations below. The lower the pH, the more acidic the solution is and the higher the pH, the less acidic. The lower the pOH, the more basic the solution is and the higher the pOH, the less basic.
Key Equations
- Ka × Kb = [H3O+][OH–] = Kw
- 14.00 = pH + pOH
- pH = -log[H3O+]
- [H3O+] = 10-pH
- pOH = -log[OH–]
- [OH–] = 10-pOH
Glossary
- acidic
- a solution where [H3O+] > [OH–]
- autoionization
- a reaction where two of the same molecule react and a proton transfers between them
- basic
- a solution where [H3O+] < [OH–]
- ion-product constant for water (Kw)
- the equilibrium constant for the autoionization of water, 2H2O(ℓ) ⇌ H3O+(aq) + OH–(aq)
- neutral
- a solution where [H3O+] = [OH–]
Chemistry End of Section Exercises
- Explain why a sample of pure water at 40 °C is neutral even though [H3O+] = 1.7 × 10−7 M. Kw is 2.9 × 10−14 at 40 °C.
- The ionization constant for water (Kw) is 2.9 × 10−15 at 10 °C. Calculate [H3O+], [OH−], pH, and pOH for pure water at 10 °C.
- The ionization constant for water (Kw) is 9.311 × 10−14 at 60 °C. Calculate [H3O+], [OH−], pH, and pOH for pure water at 60 °C.
- Calculate the pH and the pOH of each of the following solutions at 25 °C for which the substances ionize completely:
- 0.200 M HCl
- 0.0143 M NaOH
- 3.0 M HNO3
- 0.0031 M Ca(OH)2
- What are the hydronium and hydroxide ion concentrations in a solution whose pH is 6.52?
- The hydronium ion concentration in a sample of rainwater is found to be 1.7 × 10−6 M at 25 °C. What is the concentration of hydroxide ions in the rainwater?
- What is the pH of 0.21 M Ba(OH)2(aq)?
- What is the pH of 1.30 M HNO3(aq)?
- List the following in terms of increasing pH:
1.0 M HNO3(aq); 1.0 M KOH(aq); 1.0 M CH3COOH(aq); 1.0 M NH3(aq)
- Which of the following solutions are acidic? Select all that apply.
- A solution with pOH = 10.32
- A solution with [OH–] = 4.2 × 10–9 M
- A solution with [H3O+] = 1.2 × 10–9 M
- Which of the following has the highest pOH value?
- A solution with a pOH of 2.31
- A solution with a pH of 2.31
- A solution with [OH–] = 3.44 × 10–11 M
- A solution with [H3O+] = 3.44 × 10–11 M
- A solution with [OH–] = 4.24 × 10–12 M
- Given 0.10 M solutions of the following acids, which will have the highest pH?
- hydroazoic acid, Ka = 1.9 × 10–5
- formic acid, Ka = 1.8 × 10–4
- chloroacetic acid, Ka = 1.4 × 10–3
- benzoic acid, Ka = 6.3 × 10–5
- lactic acid, Ka = 1.4 × 10–4
- Anhydrous liquid ammonia, NH3(ℓ), contains no water. The NH3 undergoes autoionization analogous (in a similar fashion) to the autoionization of H2O. What chemical species, apart from NH3(ℓ), can be found in pure liquid ammonia?
Answers to Chemistry End of Section Exercises
- In a neutral solution [H3O+] = [OH−]. At 40 °C, [H3O+] = [OH−] = (2.9 × 10−14)1/2 = 1.7 × 10−7 M.
- [H3O+] = 5.4 × 10-8 M, [OH−] = 5.4 × 10-8 M, pH = 7.27, and pOH = 7.27
- [H3O+] = 3.051 × 10-7, [OH−] = 3.051 × 10-7 M, pH = 6.5156, and pOH = 6.5156
- (a) pH = 0.699, pOH = 13.301
(b) pH = 12.155, pOH = 1.845
(c) pH = -0.48, pOH = 14.48
(d) pH = 11.79, pOH = 2.21 - [H3O+] = 3.0 × 10-7 M, [OH–] = 3.3 × 10-8 M
- [OH–] = 5.9 × 10-9 M
- pH = 13.62
- pH = –0.114
- 1.0 M HNO3(aq) < 1.0 M CH3COOH(aq) < 1.0 M NH3(aq) < 1.0 M KOH(aq)
- A and B
- B
- A
- NH2– and NH4+
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

