M8Q1: Bonding and Electronegativity
Introduction
This section explores electronegativity and how it relates to bonding. We will then talk more in depth about two important types of bonding, covalent and ionic bonding. The section below provides a more detailed description of these topics, worked examples, practice problems, and a glossary of important terms.
Learning Objectives for Bonding and Electronegativity
- Apply the concept of electronegativity to characterize a bond as covalent, ionic, or polar covalent.
| Formation of Covalent Bonds | Electronegativity | Nonpolar vs. Polar Covalent Bonds | Ionic Bonds |
| Key Concepts and Summary | Glossary | End of Section Exercises |
Formation of Covalent Bonds
Nonmetal atoms frequently form covalent bonds with other nonmetal atoms. For example, the hydrogen molecule, H2, contains a covalent bond between its two hydrogen atoms. Figure 1 illustrates how this bond is formed. Starting on the far right, we have two separate hydrogen atoms with a particular potential energy, indicated by the red line. Along the x-axis is the distance between the two atoms. As the two atoms approach each other (moving left along the x-axis), their orbitals (including their valence 1s orbitals) begin to overlap. The single electrons on each hydrogen atom then interact with both atomic nuclei, occupying the space between both atoms. The system is stabilized by the two electrons occupying a lower energy bonding orbital that results from the overlap of the valence 1s orbitals. The attraction of each shared electron to both nuclei further stabilizes the system, and the potential energy decreases as the bond distance decreases. If the atoms continue to approach each other, the positive charges in the two nuclei begin to repel each other, and the potential energy increases. The bond length is determined by the distance at which the lowest potential energy is achieved.
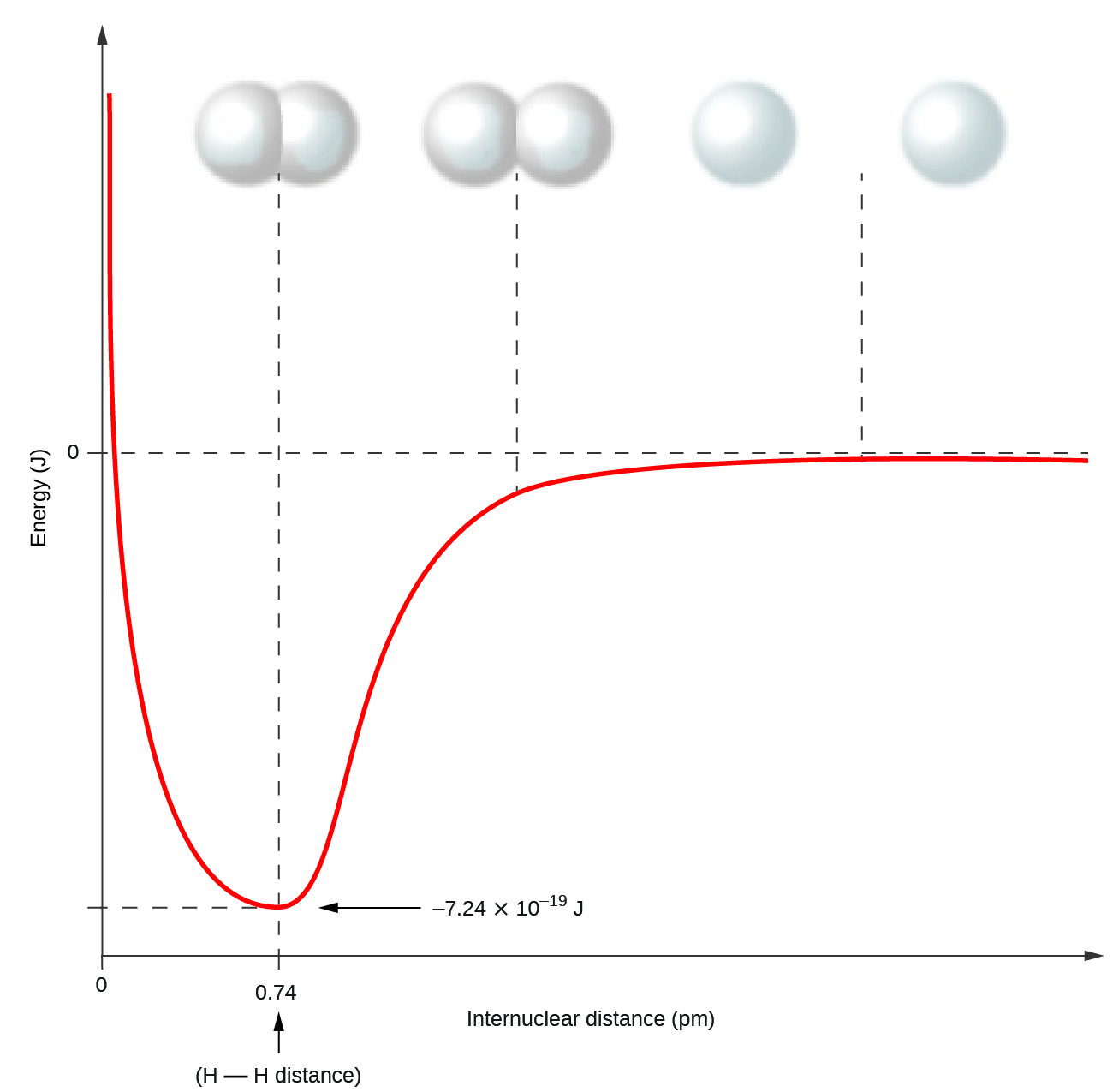
It is essential to remember that energy must be added to break chemical bonds (an endothermic process), whereas forming chemical bonds releases energy (an exothermic process). In the case of H2, the covalent bond is very strong; a large amount of energy, 436 kJ, must be added to break the bonds in one mole of hydrogen molecules and cause the atoms to separate:
H2(g) → 2 H(g) ΔH = 436 kJ/mol
Conversely, the same amount of energy is released when one mole of H2 molecules forms from two moles of H atoms:
2 H(g) → H2(g) ΔH = -436 kJ/mol
Electronegativity
Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Electronegativity is a measure of the tendency of an atom to attract electrons (or electron density) within a bond. Electronegativity differences can be used to predict how shared electrons are distributed between the two nuclei in a bond. The more strongly an atom attracts the electrons within its bonds, the larger its electronegativity value. Electrons in a polar covalent bond are shifted toward the more electronegative atom. Thus, the more electronegative atom is the one with the partial negative charge, and the less electronegative atom is the one with the partial positive charge. The greater the difference in electronegativity, the more polarized the electron distribution and the larger the partial charges of the atoms.
Figure 2 and Figure 3 show the electronegativity values of the elements as proposed by one of the most famous chemists of the twentieth century: Linus Pauling (Figure 4). (These values can also be found in Appendix H.) Other scientists have proposed different measures and values for electronegativities, so you may see slightly different values depending on the source used. In general, electronegativity increases from left to right across a period in the periodic table and decreases down a group. Thus, the nonmetals, which lie in the upper right, tend to have the highest electronegativities, with fluorine the most electronegative element of all (EN = 4.0). Metals tend to be less electronegative elements, and the group 1 metals have the lowest electronegativities. Note that noble gases are excluded from this figure because these atoms usually do not share electrons with others atoms since they have a full valence shell. (While noble gas compounds such as XeO2 do exist, they can only be formed under extreme conditions, and thus they do not fit neatly into the general model of electronegativity.)
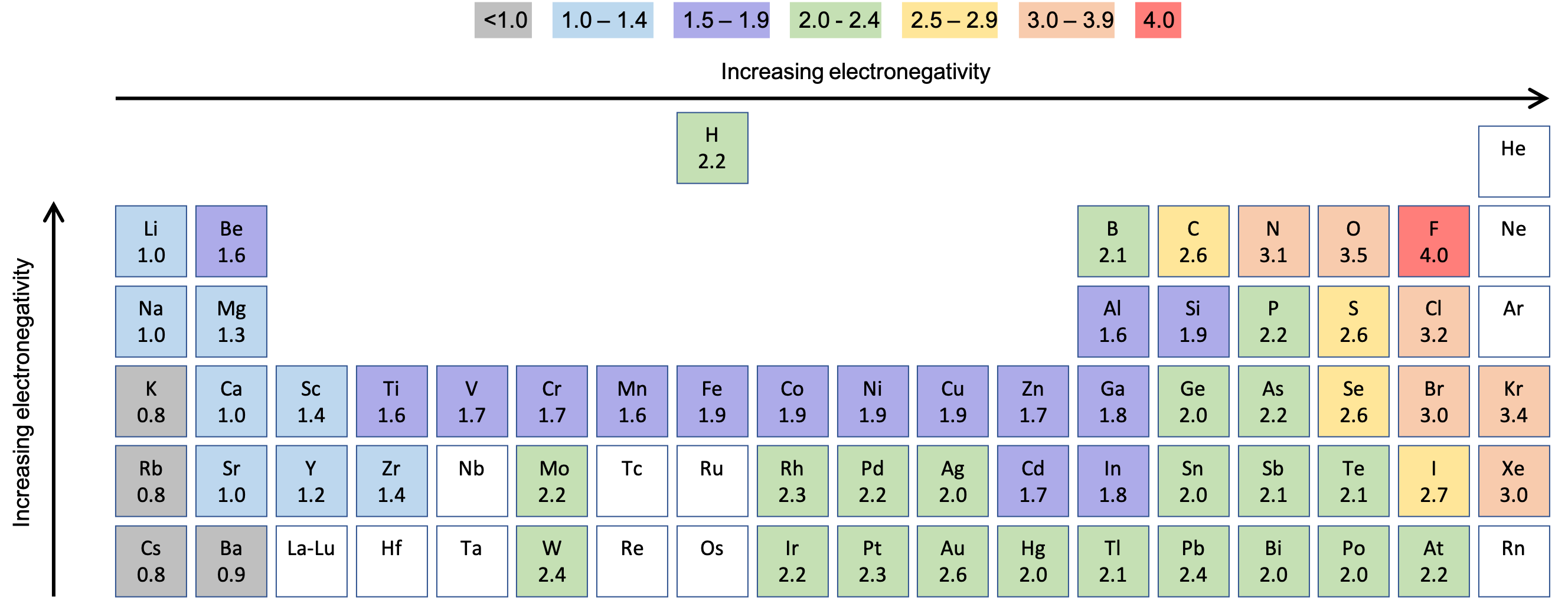
Electronegativity versus Electron Affinity
We must be careful not to confuse electronegativity and electron affinity. The electron affinity of an element is a measurable physical quantity, namely, the energy released or absorbed when an isolated gas-phase atom acquires an electron, measured in kJ/mol. Electronegativity, on the other hand, describes how tightly an atom attracts electrons in a bond. It is a dimensionless quantity that is calculated, not measured. Pauling derived the first electronegativity values by comparing the amounts of energy required to break different types of bonds. He chose an arbitrary relative scale ranging from 0 to 4.
Linus Pauling
Linus Pauling, shown in Figure 4, is the only person to have received two unshared (individual) Nobel Prizes: one for chemistry in 1954 for his work on the nature of chemical bonds and one for peace in 1962 for his opposition to weapons of mass destruction. He developed many of the theories and concepts that are foundational to our current understanding of chemistry, including electronegativity and resonance structures.

Pauling also contributed to many other fields besides chemistry. His research on sickle cell anemia revealed the cause of the disease—the presence of a genetically inherited abnormal protein in the blood—and paved the way for the field of molecular genetics. He was an unrelenting advocate for the importance of Vitamin C and human health – a very controversial opinion in his day! And his work was also pivotal in curbing the testing of nuclear weapons; he proved that radioactive fallout from nuclear testing posed a public health risk.
Electronegativity and Bond Type
The absolute value of the difference in electronegativity (ΔEN) of two bonded atoms provides a rough measure of the polarity to be expected in the bond and, thus, the bond type. When the difference is very small or zero, the bond is covalent and nonpolar. When it is large, the bond is polar covalent or ionic. Figure 5 shows the relationship between electronegativity difference and bond type.
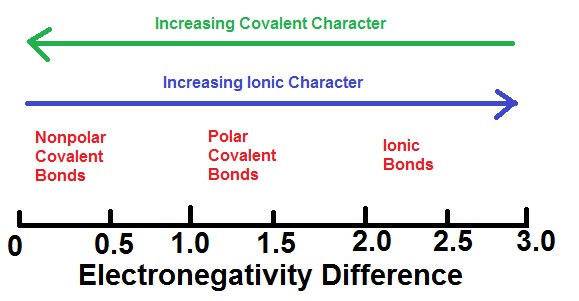
Taken together, Figure 2 and Figure 5 provide a general guide about classifying bonds, however, there are many exceptions. For example, the H and F atoms in HF have an electronegativity difference of 1.8, and the N and H atoms in NH3 a difference of 0.9, yet both of these compounds form bonds that are considered polar covalent. Likewise, the Na and Cl atoms in NaCl have an electronegativity difference of 2.2, and the Mn and I atoms in MnI2 have a difference of 1.1, yet both of these substances form ionic compounds. The best guide to the covalent or ionic character of a bond is to consider the types of atoms involved and their relative positions in the periodic table. Bonds between two nonmetals are generally covalent; bonds between a metal and a nonmetal are generally ionic.
Some compounds contain both covalent and ionic bonds. The atoms in polyatomic ions, such as OH–, NO3−, and NH4+, are held together by polar covalent bonds. However, these polyatomic ions form ionic compounds by combining with ions of opposite charge. For example, potassium nitrate, KNO3, contains the K+ cation and the polyatomic NO3− anion. Thus, bonding in potassium nitrate is ionic, resulting from the electrostatic attraction between the ions K+ and NO3−, as well as covalent between the nitrogen and oxygen atoms in NO3−.
Compounds that contain covalent bonds exhibit different physical properties than ionic compounds. The attraction between molecules is weaker than that between electrically charged ions. Thus, covalent compounds generally have much lower melting and boiling points than ionic compounds. In fact, many covalent compounds are liquids or gases at room temperature, and, in their solid states, they are typically much softer than ionic solids. We’ll explore intermolecular forces and their impact on physical properties in greater detail in Module 10.
Nonpolar vs. Polar Covalent Bonds
A nonpolar covalent bond is one in which the electrons are shared equally or nearly equally between atoms. This means that there is an equal or near-equal probability of finding the electrons near each nucleus. Consider the scenario in which the atoms that form a covalent bond are identical, as in H2 and Cl2. Since the electronegativity difference between two identical atoms is zero, the electrons in the bond must be shared equally. In the case of Cl2, each atom starts off with seven valence electrons, and each Cl shares one electron with the other, forming one covalent bond:
Cl + Cl → Cl2
The total number of electrons around each individual atom consists of six nonbonding electrons and two shared (i.e., bonding) electrons for eight total electrons, matching the number of valence electrons in the noble gas argon. This nonpolar covalent bond shares the electrons equally and there is no partial charge on either atom.
But what happens in the scenario where two different types of atoms, with different electronegativity, are bonded? In such a case, the bonding electrons would be more strongly attracted to one atom than the other, giving rise to a shift of electron density toward that atom. The extent of electron density shift can be estimated by the electronegativity difference between the two atoms. This unequal distribution of electrons gives rise to a polar covalent bond, characterized by a partial positive charge on one atom and a partial negative charge on the other. The atom that attracts the electrons more strongly acquires the partial negative charge and vice versa.
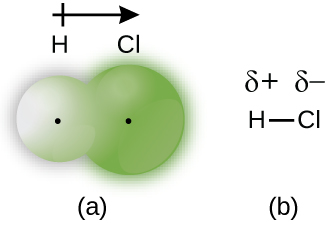
For example, the electrons in the H–Cl bond of a hydrogen chloride molecule spend more time near the chlorine atom than near the hydrogen atom. Thus, in an HCl molecule, the chlorine atom carries a partial negative charge and the hydrogen atom has a partial positive charge. Figure 6 shows the distribution of electrons in the H–Cl bond. Note that the shaded area around Cl is much larger than it is around H. Compare this to Figure 1, which shows the even distribution of electrons in the H2 nonpolar bond.
We sometimes designate the positive and negative atoms in a polar covalent bond using a lowercase Greek letter “delta,” δ, with a plus sign or minus sign to indicate whether the atom has a partial positive charge (δ+) or a partial negative charge (δ–). This symbolism is shown for the H–Cl molecule in Figure 6. We can also draw an arrow that points toward the more electronegative atom, as shown in Figure 6a and discussed more in a later section on molecule polarity.
Example 1
Electronegativity and Bond Polarity
Bond polarities play an important role in determining the structure of molecules. Use the electronegativity values in Figure 2 to designate the positive and negative atoms using the symbols δ+ and δ–:
S–H, C–N, N–H, C–O, O–H, B–H
Solution
The polarity of these bonds increases as the absolute value of the electronegativity difference increases. The atom with the δ– designation is the more electronegative of the two. Table 1 shows these bonds in order of increasing polarity.
| Bond | Polarity |
|---|---|
| S–H | |
| C–N | |
| N–H | |
| C–O | |
| O–H | |
| B–H |
Check Your Learning
Silicones are polymeric compounds containing, among others, the following types of covalent bonds: Si–O, Si–C, and C–C. Using the electronegativity values in Figure 2 to designate the positive and negative atoms using the symbols δ+ and δ–.
Answer:
| Bond | Polarity |
|---|---|
| C–C | nonpolar |
| Si–C | |
| Si–O |
Ionic Bonds
As you have learned, ions are atoms or molecules bearing an electrical charge. A cation forms when a neutral atom loses one or more electrons from its valence shell, and an anion forms when a neutral atom gains one or more electrons in its valence shell.
Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. The properties of ionic compounds shed some light on the nature of ionic bonds. Ionic solids exhibit a crystalline structure and tend to be rigid and brittle; they also tend to have high melting and boiling points, which suggests that ionic bonds are very strong. Ionic solids are also poor conductors of electricity for the same reason—the strength of ionic bonds prevents ions from moving freely in the solid state. Many ionic solids, however, dissolve readily in water. Once dissolved or melted, ionic compounds are excellent conductors of electricity (i.e., strong electrolytes) and heat because the ions can move about freely.
Neutral atoms and their associated ions have very different physical and chemical properties. Sodium atoms form sodium metal, a soft, silvery-white metal that burns vigorously in air and reacts explosively with water. Chlorine atoms form chlorine gas, Cl2, a yellow-green gas that is extremely corrosive to most metals and very poisonous to animals and plants. The vigorous reaction between the elements sodium and chlorine forms the white, crystalline compound sodium chloride, common table salt, which contains sodium cations and chloride anions (Figure 7). The compound composed of these ions exhibits properties entirely different from the properties of the elements sodium and chlorine. Chlorine is poisonous, but sodium chloride is essential to life; sodium atoms react vigorously with water, but sodium chloride simply dissolves in water.

Key Concepts and Summary
Covalent bonds form when electrons are shared between atoms and are attracted by the nuclei of both atoms. In nonpolar covalent bonds, the electrons are shared equally or nearly equally. In polar covalent bonds, the electrons are shared unequally, as one atom exerts a stronger force of attraction on the electrons than the other. The ability of an atom to attract a pair of electrons in a chemical bond is called its electronegativity. The difference in electronegativity between two atoms determines how polar a bond will be. In a diatomic molecule with two identical atoms, there is no difference in electronegativity, so the bond is nonpolar covalent. When the electronegativity difference is very large, as is the case between metals and nonmetals, the bonding is often characterized as ionic. It is not possible to ascribe strict cutoff values for the electronegativity difference that would be characterized as nonpolar covalent, polar covalent, or ionic compounds.
Atoms gain or lose electrons to form ions with particularly stable electron configurations. These ions are then held together in an ionic bond by electrostatic forces. The charges of cations formed by the representative metals may be determined readily because, with few exceptions, the electronic structures of these ions have either a noble gas configuration or a completely filled electron shell. The charges of anions formed by the nonmetals may also be readily determined because these ions form when nonmetal atoms gain enough electrons to fill their valence shells.
Glossary
- bond length
- distance between the nuclei of two bonded atoms at which the lowest potential energy is achieved
- covalent bond
- bond formed when electrons are shared between atoms
- electronegativity
- tendency of an atom to attract electrons in a bond to itself
- ionic bond
- strong electrostatic force of attraction between cations and anions in an ionic compound
- nonpolar covalent bond
- covalent bond between atoms of identical electronegativities; also C-H bond is considered close to nonpolar covalent bond
- polar covalent bond
- covalent bond between atoms of different electronegativities; a covalent bond with a positive end and a negative end
Chemistry End of Section Exercises
- Why is it incorrect to speak of solid NaCl as a molecule?
- What information can you use to predict whether a bond between two atoms is covalent or ionic?
- Predict which of the following compounds are ionic and which are covalent, based on the location of their constituent atoms in the periodic table:
- Cl2CO
- MnO
- NCl3
- CoBr2
- K2S
- CO
- CaF2
- HI
- CaO
- IBr
- CO2
- Explain the difference between a nonpolar covalent bond, a polar covalent bond, and an ionic bond.
- From its position in the periodic table, determine which atom in each pair is more electronegative:
- Br or Cl
- N or O
- S or O
- P or S
- Si or N
- Ba or P
- N or K
- From their positions in the periodic table, arrange the atoms in each of the following series in order of increasing electronegativity:
- C, F, H, N, O
- Br, Cl, F, H, I
- F, H, O, P, S
- Al, H, Na, O, P
- Ba, H, N, O, As
- Which atoms can bond to sulfur so as to produce a positive partial charge on the sulfur atom?
- Which is the most polar bond?
- C–C
- C–H
- N–H
- O–H
- Se–H
- Identify the more polar bond in each of the following pairs of bonds:
- HF or HCl
- NO or CO
- SH or OH
- PCl or SCl
- CH or NH
- SO or PO
- CN or NN
- Which of the following molecules or ions contain polar bonds? Select all that apply.
- O3
- S8
- O2
- NO3−
- CO2
- H2S
- BH4−
- Which of the following statements about oxygen and sulfur is correct?
- The sulfur 1s orbital and the oxygen 1s orbital and are the same size because both s orbitals can only have two electrons.
- The oxygen 1s orbital is smaller than the sulfur 1s orbital because oxygen is more electronegative than sulfur.
- The oxygen 1s orbital is smaller than the sulfur 1s orbital because oxygen contains fewer electrons.
- The sulfur 1s orbital is smaller than the oxygen 1s orbital because sulfur’s p and d orbitals crowd the s orbitals.
- The sulfur 1s orbital is smaller than the oxygen 1s orbital because sulfur’s nuclear charge draws the electrons closer.
Answers to Chemistry End of Section Exercises
- NaCl consists of discrete ions arranged in a crystal lattice, not covalently bonded molecules.
- The difference in electronegativity between the atoms. If the two atoms have similiar electronegativity values, they tend to form covalent bond. If the difference in electronegativity is large, the bond tends to be ionic.
- Ionic: (b), (d), (e), (g), and (i)
Covalent: (a), (c), (f), (h), (j), and (k) - Ionic bond: strong electrostatic force of attraction between cations and anions in an ionic compound
Nonpolar covalent bond: covalent bond between atoms of identical (or nearly identical) electronegativity
Polar covalent bond: covalent bond between atoms with different electronegativity; a covalent bond with a partial positive end and a partial negative end - (a) Cl; (b) O; (c) O; (d) S; (e) N; (f) P; (g) N
- (a) H < C < N < O < F
(b) H < I < Br < Cl < F
(c) H < P < S < O < F
(d) Na < Al < H < P < O
(e) Ba < H < As < N < O - N, O, F, and Cl
- D
- (a) HF; (b) CO; (c) OH; (d) PCl; (e) NH; (f) PO; (g) CN
- D, E, F, and G
- E
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

