M13Q7: Nanoscale View of Chemical Kinetics: KMT Considerations, Activation Energy, Orientation of Molecules
Learning Objectives
- Describe from a submicroscopic viewpoint why the following affect reaction rates: temperature (including interpretation of Maxwell-Boltzmann distributions), concentration, surface area, phase, and catalysts.
- Distinguish between and describe differences of unimolecular and bimolecular reactions.
- Draw and interpret reaction coordinate diagrams, and identify the transition state(s) on these diagrams.
| Key Concepts and Summary | Glossary | End of Section Exercises |
Chemical Kinetics: A Nanoscale Approach
We should not be surprised that atoms, molecules, or ions must collide before they can react with each other. Atoms must be close together to form chemical bonds. This simple premise is the basis for a very powerful theory that explains many observations regarding chemical kinetics, including factors affecting reaction rates.
The Principles of Rate Theory, also know as Collision theory, is based on the following postulates:
- Most reactions require an activation energy, Ea. Reactants must have enough energy to collide with one another, rearrange bonds, and overcome steric hindrance and other reaction-dependent parameters. The sum of these three energies is the activation energy.
- Reactants have to go through a high-energy point (transition state) before being converted to products.
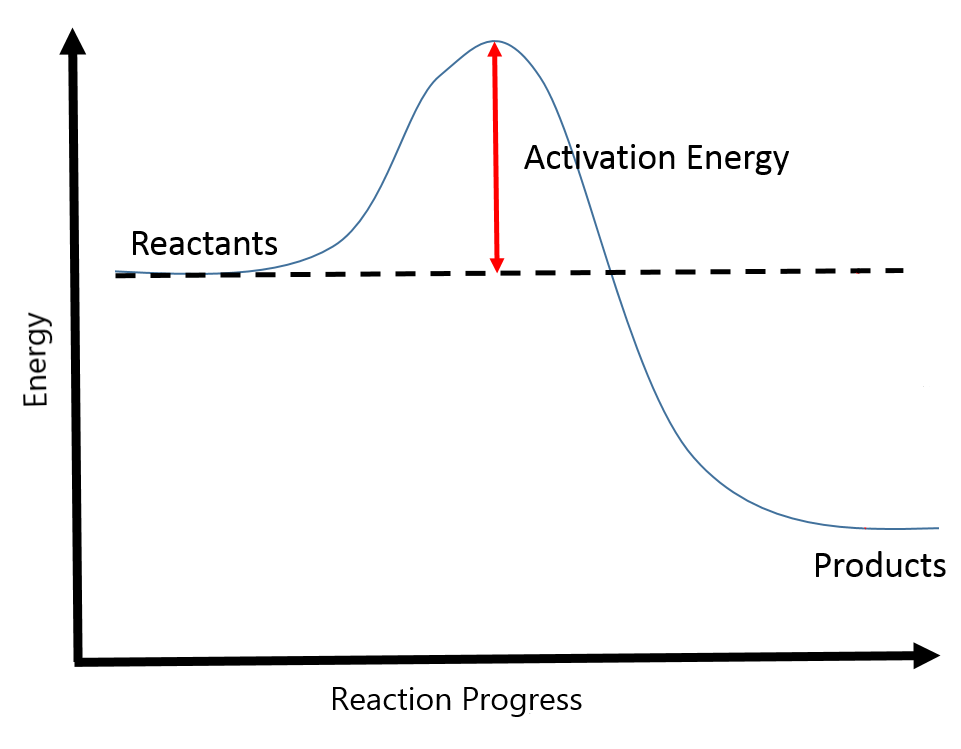
- The pathway that connects reactants to products is called the reaction coordinate. This can be seen graphically in a plot of energy versus reaction progress, as in Figure 1.
We can see the importance of the two physical factors noted in postulates 1 and 2, the orientation and energy of collisions, when we consider the reaction of carbon monoxide with oxygen:
2 CO(g) + O2(g) → 2 CO2(g)
Carbon monoxide is a pollutant produced by the combustion of hydrocarbon fuels. To reduce this pollutant, automobiles have catalytic converters that use a catalyst to carry out this reaction. It is also a side reaction of the combustion of gunpowder that results in muzzle flash for many firearms. If carbon monoxide and oxygen are present in sufficient quantity, the reaction is spontaneous at high temperature and pressure.
The first step in the gas-phase reaction between carbon monoxide and oxygen is a collision between the two molecules:
CO(g) + O2(g) → CO2(g) + O(g)
Although there are many different possible orientations the two molecules can have relative to each other, consider the two presented in Figure 2. In the first case, the oxygen side of the carbon monoxide molecule collides with the oxygen molecule. In the second case, the carbon side of the carbon monoxide molecule collides with the oxygen molecule. The second case is clearly more likely to result in the formation of carbon dioxide, which has a central carbon atom bonded to two oxygen atoms (O = C = O). This is a rather simple example of how important the orientation of the collision is in terms of creating the desired product of the reaction.
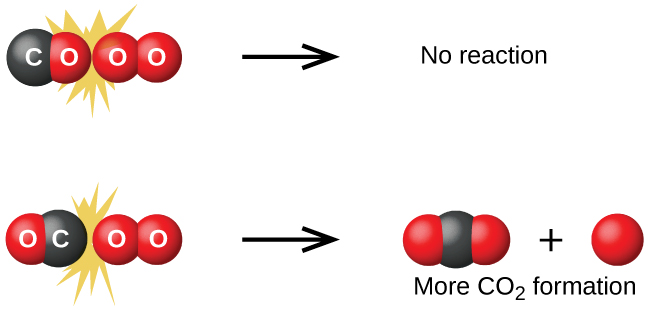
If the collision does take place with the correct orientation, there is still no guarantee that the reaction will proceed to form carbon dioxide. Every reaction requires a certain amount of activation energy for it to proceed in the forward direction, yielding an appropriate activated complex along the way. As Figure 3 demonstrates, even a collision with the correct orientation can fail to form the reaction product. In the study of reaction mechanisms, each of these three arrangements of atoms is called a transition state or activated complex.
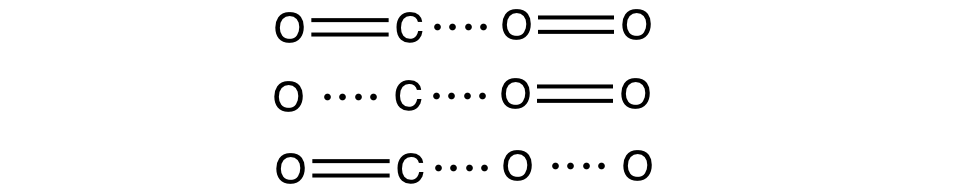
In most circumstances, it is impossible to isolate or identify a transition state or activated complex. In the reaction between carbon monoxide and oxygen to form carbon dioxide, activated complexes have only been observed spectroscopically in systems that utilize a heterogeneous catalyst. The gas-phase reaction occurs too rapidly to isolate any such chemical compound.
Collision theory explains why most reaction rates increase as concentrations increase. With an increase in the concentration of any reacting substance, the chances for collisions between molecules are increased because there are more molecules per unit of volume. More collisions mean a faster reaction rate, assuming the energy of the collisions is adequate.
Activation Energy
The minimum energy necessary to form a product during a collision between reactants is called the activation energy (Ea). The kinetic energy of reactant molecules plays an important role in a reaction because the energy necessary to form a product is provided by a collision of a reactant molecule with another reactant molecule. (In single-reactant reactions, activation energy may be provided by a collision of the reactant molecule with the wall of the reaction vessel or with molecules of an inert contaminant.) If the activation energy is much larger than the average kinetic energy of the molecules, the reaction will occur slowly: only a few fast-moving molecules will have enough energy to react. If the activation energy is much smaller than the average kinetic energy of the molecules, the fraction of molecules possessing the necessary kinetic energy will be large; most collisions between molecules will result in reaction, and the reaction will occur rapidly.
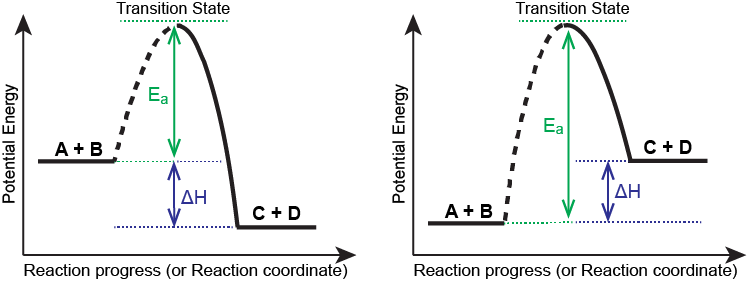
Figure 4 shows the energy relationships for the general exothermic and endothermic reactions of molecule A with molecule B to form molecules C and D:
A + B → C + D
The exothermic figure (left) shows that the energy of the transition state is higher than that of the reactants A and B by an amount equal to Ea, the activation energy. Thus, the sum of the kinetic energies of A and B must be equal to or greater than Ea to reach the transition state. After the transition state has been reached, and as C and D begin to form, the system loses energy until its total energy is lower than that of the initial mixture. This lost energy is transferred to other molecules, giving them enough energy to reach the transition state. The forward reaction (that between molecules A and B) therefore tends to take place readily once the reaction has started. In Figure 4, ΔH represents the difference in enthalpy between the reactants (A and B) and the products (C and D). The sum of Ea and ΔH represents the activation energy for the reverse reaction: C + D → A + B.
In the endothermic figure (right), the primary difference is that after the transition state has been reached and as C and D begin to form, the system loses energy until its total energy is higher than that of the initial mixture. There is no excess or lost energy to be transferred to other molecules, so energy must come from an external source for the reaction to continue. The difference between Ea and ΔH represents the activation energy for the reverse reaction: C + D → A + B.
Key Concepts and Summary
Understanding a chemical reaction at the molecular level gives us great insight into the factors that affect the reaction rate and the ability to speed up, slow down, or stop a reaction altogether. This animation of the reaction is based on Collision Theory, which generalizes reactions as having an activation energy barrier to overcome and passing through a transition state before being converted to products, all while traveling along a reaction coordinate. In this course, we are considering the reaction coordinate to be two-dimensional, but in real life, it is anything but! In Figure 4 of this section, a reaction coordinate diagram can be seen for both an exothermic (left) and endothermic (right) reaction. In both cases, the activation energy is the energy between the reactants and the transition state. The enthalpy of the reaction is the energy difference between the reactants and products.
Glossary
- activation energy (Ea)
- the minimum energy necessary to form a product during a collision between reactants
- collision theory (principles of rate theory)
- a theory that explains many observations regarding chemical kinetics, based on the postulates that most reactions require an activation energy, reactants have to go through a transition state, and every reaction has a reaction coordinate.
- reaction coordinate
- the pathway that connects reactants to products
- transition state
- a high-energy point between reactants, intermediates, or products.
Chemistry End of Section Exercises
- In terms of collision theory, to which of the following is the rate of a chemical reaction proportional?
- the change in free energy per second
- the change in temperature per second
- the number of collisions per second
- the number of product molecules
- Chemical reactions occur when reactants collide. What are two factors that may prevent a collision from producing a chemical reaction?
- What is the activation energy of a reaction, and how is this energy related to the activated complex of the reaction?
- A Maxwell-Boltzmann Distribution plot shows the speeds for gas molecules at a certain temperatures. Use the plot below, depicting a reactant at various temperatures, to answer the following questions:
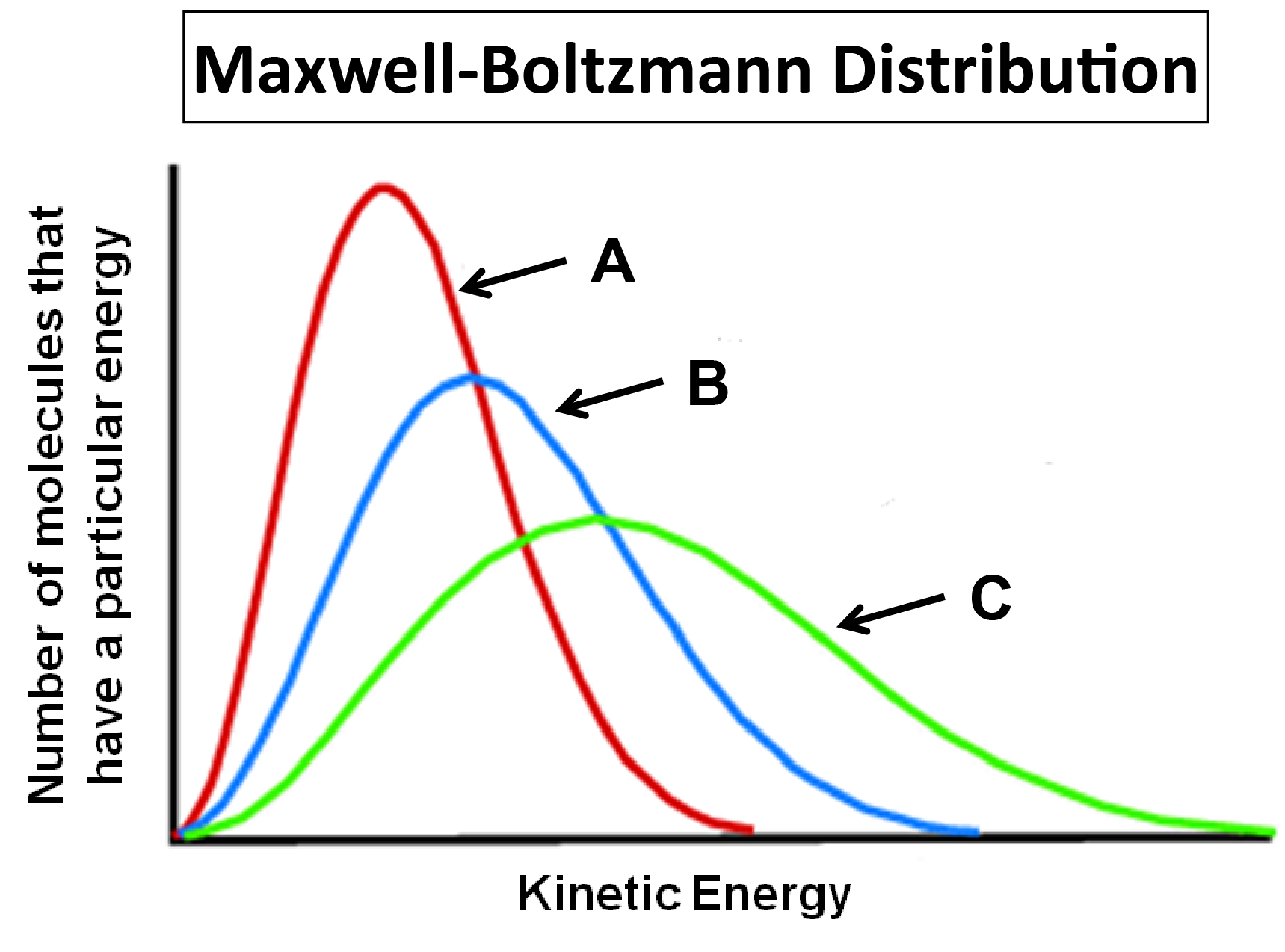
- Which distribution represents the reactant at the lowest temperature?
- Which distribution represents the fastest reaction? And why?
- The combustion reaction of propane and oxygen is a highly exothermic reaction, however, it is possible to keep propane and oxygen together without any observable reaction. Why is this?
Answers to Chemistry End of Section Exercises
- B and C
- The reactants either may be moving too slowly to have enough kinetic energy to exceed the activation energy for the reaction, or the orientation of the molecules when they collide may prevent the reaction from occurring.
- The activation energy is the minimum amount of energy necessary to form the activated complex in a reaction. It is usually expressed as the energy necessary to form one mole of activated complex.
- (a) A; (b) C, The reactant shown in curve C has the highest average kinetic energy, meaning more reactants will collide with enough energy to overcome the reaction’s activation energy.
- In order to react, reactant must overcome the reaction’s activation energy. At room temperature, propane and oxygen molecules do not not possess a high enough average kinetic energy to overcome the activation energy.
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

