M14Q5: Calculations Involving the Equilibrium Constant
Learning Objectives
- Determine the value of an equilibrium constant from equilibrium concentrations.
- Calculate the concentration of a reactant or product at equilibrium when given the value of the equilibrium constant.
| Key Concepts and Summary | Glossary | End of Section Exercises |
While we have learned to identify which direction a reaction will shift to reach equilibrium, we want to extend that understanding to quantitative calculations. We do so by evaluating the ways that the concentrations of products and reactants change as a reaction approaches equilibrium, keeping in mind the stoichiometric coefficients within the reaction. This algebraic approach to equilibrium calculations will be explored in this section.
Evaluating Stoichiometric Coefficients
Changes in concentrations or pressures of reactants and products occur as a reaction system approaches equilibrium. In this section, we will see that we can relate these changes to each other using the coefficients in the balanced chemical equation describing the system. We use the decomposition of ammonia as an example.
On heating, ammonia reversibly decomposes into nitrogen and hydrogen gases according to this equation:
2 NH3(g) ⇌ N2(g) + 3 H2(g)
If a sample of ammonia decomposes in a closed system and the concentration of N2 increases by 0.11 M, the change in the N2 concentration is 0.11 M. We can define the change in concentration by Δ[N2], the final concentration minus the initial concentration of N2. The change is positive because the concentration of N2 increases.
The change in the H2 concentration, Δ[H2], is also positive—the concentration of H2 increases as ammonia decomposes. The chemical equation tells us that the change in the concentration of H2 is three times the change in the concentration of N2 because for each mole of N2 produced, 3 moles of H2 are produced.
Δ[H2] = 3 × Δ[N2] = 3 × (0.11 M) = 0.33 M
The change in concentration of NH3, Δ[NH3], is twice that of Δ[N2]; the equation indicates that 2 moles of NH3 must decompose for each mole of N2 formed. However, the change in the NH3 concentration is negative because the concentration of ammonia decreases as it decomposes.
Δ[NH3] = -2 × Δ[N2] = -2 × (0.11 M) = -0.22 M
If we did not know the magnitude of the change in the concentration of N2, we could represent it by the variable, x. Δ[N2] = +x
The coefficients in the Δ terms are identical to those in the balanced equation for the reaction.
2 NH3(g) ⇌ N2(g) + 3 H2(g)
-2x +x +3x
Note that all the changes on one side of the arrows are of the same sign and that all the changes on the other side of the arrows are of the opposite sign. The simplest way for us to find the coefficients for the concentration changes in any reaction is to use the coefficients in the balanced chemical equation.
Example 1
Determining Relative Changes in Concentration
Complete the changes in concentrations for each of the following reactions.
(a) C2H2(g) + 2 Br2(g) ⇌ C2H2Br4(g)
(b) I2(aq) + I–(aq) ⇌ I3–(aq)
(c) C3H8(g) + 5 O2(g) ⇌ 3 CO2(g) + 4 H2O(g)
Solution
(a) C2H2(g) + 2 Br2(g) ⇌ C2H2Br4(g)
(b) I2(aq) + I–(aq) ⇌ I3–(aq)
(c) C3H8(g) + 5 O2(g) ⇌ 3 CO2(g) + 4 H2O(g)
Check Your Learning
Complete the changes in concentrations for each of the following reactions:
(a) 2 SO2(g) + O2(g) ⇌ 2 SO3(g)
(b) C4H8(g) ⇌ 2 C2H4(g)
(c) 4 NH3(g) + 7 H2O(g) ⇌ 4 NO2(g) + 6 H2O(g)
Answer:
(a) +2x, +x, −2x
(b) +x, −2x
(c) +4x, +7x, −4x, −6x; or −4x, −7x, +4x, +6x
Calculation of an Equilibrium Constant
Any problem that requires us to solve for Kc must provide enough information to determine the reactant and product concentrations at equilibrium. Armed with the concentrations, we can solve the equation for Kc, as it will be the only unknown.
The following example shows how to use the stoichiometry of a reaction and a combination of initial and equilibrium concentrations to determine an equilibrium constant. This technique, commonly called an ICE chart—for Initial, Change, and Equilibrium–will be helpful in solving many equilibrium problems. A chart is generated beginning with the equilibrium reaction in question. Underneath the reaction the initial concentrations of the reactants and products are listed—these conditions are usually provided in the problem and we consider no shift toward equilibrium to have happened. The next row of data is the change that occurs as the system shifts toward equilibrium—do not forget to consider the reaction stoichiometry as described in a previous section of this chapter. The last row contains the concentrations once equilibrium has been reached.
Example 2
Calculation of an Equilibrium Constant
Iodine molecules react reversibly with iodide ions to produce triiodide ions.
I2(aq) + I–(aq) ⇌ I3–(aq)
If a solution with the concentrations of I2 and I− both equal to 1.000 × 10−3 M before reaction gives an equilibrium concentration of I2 of 6.61 × 10−4 M, what is the equilibrium constant for the reaction?
Solution
We will begin this problem by calculating the changes in concentration as the system goes to equilibrium. Then we determine the equilibrium concentrations and, finally, the equilibrium constant. First, we set up a table with the initial concentrations, the changes in concentrations, and the equilibrium concentrations using −x as the change in concentration of I2.
| I2(aq) + I–(aq) ⇌ I3–(aq) | |||
| I: Initial concentration (M) | 1.000 × 10-3 | 1.000 × 10-3 | 0 |
| C: Change (M) | –x | –x | +x |
| E: Equilibrium concentration (M) | 1.000 × 10-3 – x | 1.000 × 10-3 – x | x |
Since the equilibrium concentration of I2 is given, we can solve for x. At equilibrium the concentration of I2 is 6.61 × 10−4M so that
1.000 × 10-3 – x = 6.61 × 10-4
x = 3.39 × 10-4 M
Now we can fill in the table with the concentrations at equilibrium.
| I2(aq) + I–(aq) ⇌ I3–(aq) | |||
| I: Initial concentration (M) | 1.000 × 10-3 | 1.000 × 10-3 | 0 |
| C: Change (M) | -3.39 × 10-4 | -3.39 × 10-4 | +3.39 × 10-4 |
| E: Equilibrium concentration (M) | 6.61 × 10-4 | 6.61 × 10-4 | 3.39 × 10-4 |
We now calculate the value of the equilibrium constant.
Kc = ![]() = 776
= 776
Check Your Learning
Ethanol and acetic acid react and form water and ethyl acetate, the solvent responsible for the odor of some nail polish removers.
C2H5OH + CH3CO2H ⇌ CH3CO2C2H5 + H2O
When 1 mol each of C2H5OH and CH3CO2H are allowed to react in 1 L of the solvent dioxane, equilibrium is established when 0.33 mol of each of the reactants remains. Calculate the equilibrium constant for the reaction. (Note: Water is not a solvent in this reaction. This time, it is a reactant dissolved in an organic solvent and therefore should be included in the equilibrium expression.)
Answer:
Kc = 4.1
Calculation of a Missing Equilibrium Concentration
If we know the equilibrium constant for a reaction and know the concentrations at equilibrium of all reactants and products except one, we can calculate the missing concentration.
Example 3
Calculation of a Missing Equilibrium Concentration
Nitrogen oxides are air pollutants produced by the reaction of nitrogen and oxygen at high temperatures. At 2000 °C, the value of the equilibrium constant for the reaction, N2(g) + O2(g) ⇌ 2 NO(g), is 4.1 × 10−4. Find the concentration of NO(g) in an equilibrium mixture with air at 1 atm pressure at this temperature. In air, [N2] = 0.036 mol/L and [O2] 0.0089 mol/L.
Solution
We are given all of the equilibrium concentrations except that of NO. Thus, we can solve for the missing equilibrium concentration by rearranging the equation for the equilibrium constant.
Kc = ![]()
[NO]2 = Kc[N2][O2]
[NO] = ![]() = 3.6 × 10-4
= 3.6 × 10-4
Thus [NO] is 3.6 × 10−4 mol/L at equilibrium under these conditions.
We can check our answer by substituting all equilibrium concentrations into the expression for the reaction quotient to see whether it is equal to the equilibrium constant.
Qc = ![]() = 4.0 × 10-4 = Kc
= 4.0 × 10-4 = Kc
The answer checks; our calculated value gives the equilibrium constant within the error associated with the significant figures in the problem.
Check Your Learning
The equilibrium constant for the reaction of nitrogen and hydrogen to produce ammonia at a certain temperature is 6.00 × 10−2. Calculate the equilibrium concentration of ammonia if the equilibrium concentrations of nitrogen and hydrogen are 4.26 M and 2.09 M, respectively.
Answer:
[NH3] = 1.53 M
Calculation of Changes in Concentration
If we know the equilibrium constant for a reaction and a set of concentrations of reactants and products that are not at equilibrium, we can calculate the changes in concentrations as the system comes to equilibrium, as well as the new concentrations at equilibrium. The typical procedure can be summarized in four steps.
- Determine the direction the reaction proceeds to come to equilibrium.
- Write a balanced chemical equation for the reaction.
- If the direction in which the reaction must proceed to reach equilibrium is not obvious, calculate Qc from the initial concentrations and compare to Kc to determine the direction of change.
- Determine the relative changes needed to reach equilibrium, then write the equilibrium concentrations in terms of these changes.
- Define the changes in the initial concentrations that are needed for the reaction to reach equilibrium. Generally, we represent the smallest change with the symbol x and express the other changes in terms of the smallest change.
- Define missing equilibrium concentrations in terms of the initial concentrations and the changes in concentration determined in (a).
- Solve for the change and the equilibrium concentrations.
- Substitute the equilibrium concentrations into the equilibrium constant expression, solve for x, and check any assumptions used to find x.
- Calculate the equilibrium concentrations.
- Check the arithmetic.
- Check the calculated equilibrium concentrations by substituting them into the equilibrium expression and determining whether they give the equilibrium constant.
Sometimes a particular step may differ from problem to problem—it may be more complex in some problems and less complex in others. However, every calculation of equilibrium concentrations from a set of initial concentrations will involve these steps. In solving equilibrium problems that involve changes in concentration, sometimes it is convenient to set up an ICE table, as described in the previous section.
Example 4
Calculation of Concentration Changes as a Reaction Goes to Equilibrium
Under certain conditions, the equilibrium constant for the decomposition of PCl5(g) into PCl3(g) and Cl2(g) is 0.0211. What are the equilibrium concentrations of PCl5, PCl3, and Cl2 if the initial concentration of PCl5 was 1.00 M?
Solution
Use the stepwise process described earlier.
- Determine the direction the reaction proceeds.
The balanced equation for the decomposition of PCl5 is
PCl5(g) ⇌ PCl3(g) + Cl2(g)Because we have no products initially, Qc = 0 and the reaction will proceed to the right.
- Determine the relative changes needed to reach equilibrium, then write the equilibrium concentrations in terms of these changes.
Let us represent the increase in concentration of PCl3 by the symbol x. The other changes may be written in terms of x by considering the coefficients in the chemical equation.
PCl5(g) ⇌ PCl3(g) + Cl2(g)
-x +x +xThe changes in concentration and the expressions for the equilibrium concentrations are:
PCl5(g) ⇌ PCl3(g) + Cl2(g) I: Initial concentration (M) 1.00 0 0 C: Change (M) –x +x +x E: Equilibrium concentration (M) 1.00 – x x x - Solve for the change and the equilibrium concentrations.
Substituting the equilibrium concentrations into the equilibrium constant equation gives
Kc =
![Rendered by QuickLaTeX.com \dfrac{[\text{PCl}_{3}][\text{Cl}_{2}]}{[\text{PCl}_{5}]}](https://wisc.pb.unizin.org/app/uploads/quicklatex/quicklatex.com-47a9ffb552d7a4d23a59122396221a6c_l3.png) = 0.0211 =
= 0.0211 = 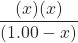
This equation contains only one variable, x, the change in concentration. We can write the equation as a quadratic equation and solve for x using the quadratic formula.
0.0211 =
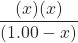
0.0211(1.00 – x) = x2
x2 + 0.0211x – 0.0211 = 0Appendix B shows us an equation of the form ax2 + bx + c = 0 can be rearranged to solve for x:
x =
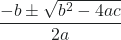
In this case, a = 1, b = 0.0211, and c = −0.0211. Substituting the appropriate values for a, b, and c yields:
x =
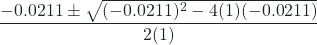
x =
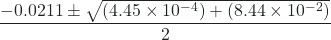
x =
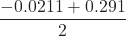 = 0.135, or, x =
= 0.135, or, x = 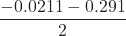 = -0.156
= -0.156Quadratic equations generally have two different solutions, one that is physically possible and one that is physically impossible (an extraneous root). In this case, the second solution (−0.156) is physically impossible because we know the change must be a positive number (otherwise we would end up with negative values for concentrations of the products). Thus, x = 0.135 M.
The equilibrium concentrations are
[PCl5] = 1.00 – 0.135 = 0.87 M
[PCl3] = x = 0.135 M
[Cl2] = x = 0.135 M - Check the arithmetic.
Substitution into the expression for Kc (to check the calculation) gives
Kc =
![Rendered by QuickLaTeX.com \dfrac{[\text{PCl}_{3}][\text{Cl}_{2}]}{[\text{PCl}_{5}]} = \dfrac{(0.135)(0.135)}{0.87}](https://wisc.pb.unizin.org/app/uploads/quicklatex/quicklatex.com-955f9b6d8a72494295cdbd484c7fb58f_l3.png) = 0.021
= 0.021The equilibrium constant calculated from the equilibrium concentrations is equal to the value of Kc given in the problem (when rounded to the proper number of significant figures). Thus, the calculated equilibrium concentrations check.
Check Your Learning
Acetic acid, CH3CO2H, reacts with ethanol, C2H5OH, to form water and ethyl acetate, CH3CO2C2H5.
CH3CO2H + C2H5OH ⇌ CH3CO2C2H5 + H2O
The equilibrium constant for this reaction with dioxane as a solvent is 4.0 (Note: Water is not a solvent in this reaction. This time it is a reactant dissolved in an organic solvent and therefore should be included in the equilibrium expression). What are the equilibrium concentrations when a mixture that is 0.15 M in CH3CO2H, 0.15 M in C2H5OH, 0.40 M in CH3CO2C2H5, and 0.40 M in H2O are mixed in enough dioxane to make 1.0 L of solution?
Answer:
[CH3CO2H] = 0.18 M, [C2H5OH] = 0.18 M, [CH3CO2C2H5] = 0.37 M, [H2O] = 0.37 M
Check Your Learning
A 1.00-L flask is filled with 1.00 moles of H2 and 2.00 moles of I2. The value of the equilibrium constant for the reaction of hydrogen and iodine reacting to form hydrogen iodide is 50.5 under the given conditions. What are the equilibrium concentrations of H2, I2, and HI in moles/L?
H2(g) + I2(g) ⇌ 2 HI(g)
Answer:
[H2] = 0.06 M, [I2] = 1.06 M, [HI] = 1.88 M
Sometimes it is possible to use chemical insight to find solutions to equilibrium problems without actually solving a quadratic (or more complicated) equation. First, however, it is useful to verify that equilibrium can be obtained starting from two extremes: all (or mostly) reactants and all (or mostly) products.
Consider the ionization of 0.150 M HA, a weak acid.
HA(aq) + H2O(ℓ) ⇌ H3O+(aq) + A–(aq) Kc = 6.80 × 10-4
The most obvious way to determine the equilibrium concentrations would be to start with only reactants. This could be called the “all reactant” starting point. Using x for the amount of acid ionized at equilibrium, this is the ICE table and solution.
| HA(aq) + H2O(ℓ) ⇌ H3O+(aq) + A–(aq) | ||||
| I: Initial concentration (M) | 0.150 | – | 0 | 0 |
| C: Change (M) | –x | – | +x | +x |
| E: Equilibrium Concentration (M) | 0.150 – x | – | x | x |
Setting up and solving the quadratic equation gives
Kc = ![]() = 6.80 × 10-4
= 6.80 × 10-4
x2 + (6.80 × 10-4)(x) – 1.02 × 10-4 = 0
x = ![]() = 0.00977 M or -0.0104 M
= 0.00977 M or -0.0104 M
Using the positive (physical) root, the equilibrium concentrations are
[HA] = 0.150 – x = 0.140 M
[H3O+] = [A–] = x = 0.00977 M
Recall that a small Kc means that very little of the reactants form products and a large Kc means that most of the reactants form products. If the system can be arranged so it starts “close” to equilibrium, then if the change (x) is small compared to any initial concentrations, it can be neglected. Small is usually defined as resulting in an error of less than 5%. The following two examples demonstrate this.
Example 5
Approximate Solution Starting Close to Equilibrium
What are the concentrations at equilibrium of a 0.15 M solution of HCN?
HCN(aq) + H2O(ℓ) ⇌ H3O+(aq) + CN–(aq) Kc = 4.9 × 10-10
Solution
Using “x” to represent the concentration of each product at equilibrium gives this ICE table.
| HCN(aq) + H2O(ℓ) ⇌ H3O+(aq) + CN–(aq) | ||||
| I: Initial concentration (M) | 0.15 | – | 0 | 0 |
| C: Change (M) | –x | – | +x | +x |
| E: Equilibrium Concentration (M) | 0.15 – x | – | x | x |
The exact solution may be obtained using the quadratic formula with
Kc = ![]()
solving
x2 + (4.9 × 10-10)x – (7.35 × 10-11) = 0
x = 8.6 × 10-6 M
Thus [H3O+] = [CN–] = x = 8.6 × 10–6 M and [HCN] = 0.15 – x = 0.15 M.
In this case, chemical intuition can provide a simpler solution. From the equilibrium constant and the initial conditions, x must be small compared to 0.15 M. More formally, if x << 0.15, then 0.15 – x ≈ 0.15. If this assumption is true, then it simplifies obtaining x
Kc = ![]() ≈
≈ ![]()
4.9 × 10-10 = ![]()
x2 = (0.15)(4.9 × 10-10) = 7.4 × 10-11
x = ![]() = 8.6 × 10–6 M
= 8.6 × 10–6 M
In this example, solving the exact (quadratic) equation and using approximations gave the same result to two significant figures. While most of the time the approximation is a bit different from the exact solution, as long as the error is less than 5%, the approximate solution is considered valid. In this problem, the 5% applies to IF (0.15 – x) ≈ 0.15 M, so if
![]() × 100% =
× 100% = ![]() × 100% = 0.006%
× 100% = 0.006%
is less than 5%, as it is in this case, the assumption is valid. The approximate solution is thus a valid solution.
Check Your Learning
What are the equilibrium concentrations in a 0.25 M NH3 solution?
NH3(aq) + H2O(ℓ) ⇌ NH4+(aq) + OH–(aq) Kc = 1.8 × 10-5
Assume that x is much less than 0.25 M and calculate the error in your assumption.
Answer:
[OH–] = [NH4+] = 0.0021 M; [NH3] = 0.25 M, error = 0.84%
The second example requires that the original information be processed a bit, but it still can be solved using a small x approximation.
Example 6
Approximate Solution After Shifting Starting Concentration
Copper(II) ions form a complex ion in the presence of ammonia
Cu2+(aq) + 4 NH3(aq) ⇌ Cu(NH3)42+(aq) Kc = 5.0 × 1013 = ![]()
If 0.010 mol Cu2+ is added to 1.00 L of a solution that is 1.00 M NH3 what are the concentrations when the system comes to equilibrium?
Solution
The initial concentration of copper(II) is 0.010 M. The equilibrium constant is very large so it would be better to start with as much product as possible because “all products” is much closer to equilibrium than “all reactants.” Note that Cu2+ is the limiting reactant; if all 0.010 M of it reacts to form product the concentrations would be
[Cu2+] = 0.010 – 0.010 = 0 M
[Cu(NH3)42+] = 0.010 M
[NH3] = 1.00 – (4 × 0.010) = 0.96 M
Using these “shifted” values as initial concentrations with x as the free copper(II) ion concentration at equilibrium gives this ICE table.
| Cu2+(aq) + 4 NH3(aq) ⇌ Cu(NH3)42+(aq) | |||
| I: Initial concentration (M) | 0 | 0.96 | 0.010 |
| C: Change (M) | +x | +4x | -x |
| E: Equilibrium concentration (M) | + x | 0.96 + 4x | 0.010 – x |
Since we are starting close to equilibrium, x should be small so that
0.96 + 4x ≈ 0.96 M
0.010 – x ≈ 0.010 M
Kc = ![]() ≈
≈ ![]() = 5.0 × 1013
= 5.0 × 1013
x = ![]() = 2.4 × 10-16 M
= 2.4 × 10-16 M
Select the smallest concentration for the 5% rule.
![]() × 100% = 2 × 10-12%
× 100% = 2 × 10-12%
This is much less than 5%, so the assumptions are valid. The concentrations at equilibrium are
[Cu2+] = x = 2.4 × 10-16 M
[NH3] = 0.96 – 4x = 0.96 M
[Cu(NH3)42+] = 0.010 – x = 0.010 M
By starting with the maximum amount of product, this system was near equilibrium and the change (x) was very small. With only a small change required to get to equilibrium, the equation for x was greatly simplified and gave a valid result well within the 5% error maximum.
Check Your Learning
What are the equilibrium concentrations when 0.25 mol Ni2+ is added to 1.00 L of 2.00 M NH3 solution?
Ni2+(aq) + 6 NH3(aq) ⇌ Ni(NH3)62+(aq) Kc = 5.5 × 108
With such a large equilibrium constant, first form as much product as possible, then assume that only a small amount (x) of the product shifts left. Calculate the error in your assumption.
Answer:
[Ni(NH3)62+] = 0.25 M, [NH3] = 0.50 M, [Ni2+] = 2.9 × 10–8M, error = 1.2 × 10–5%
Key Concepts and Summary
In this section, we dive into the types of calculations that are common in systems at equilibrium. Most of these types of questions require you to first write the chemical reaction, then set up an ICE chart, then setting up at Keq expression using the values in “E” of the ICE chart. In these types of problems, there are three primary variables – the value of Keq, the initial concentrations, and the equilibrium concentrations. Most often, two of these variables are measured and the third is unknown and needs to be calculated. If a system is not initially at equilibrium, we can calculate Q and when compared to Keq, we know which way the reaction will shift in order to achieve equilibrium. This shift is reflected in “C” of the ICE chart – the side the equilibrium shifts to contains the “+x“.
Glossary
- ICE chart
- Initial, Change, Equilibrium – a chart helpful in solving many equilibrium problems
Chemistry End of Section Exercises
- The decomposition of bromine pentafluoride can be described by the following equilibrium:
2 BrF5(g) ⇌ Br2(g) + 5 F2(g) Kc = 1.7 × 10-11
If the initial concentration of BrF5 in a sealed flask is 0.10 M, what algebraic equation would need to be solved to determine the equilibrium concentrations of all three chemical species?
- NH4CO2NH2(s) is placed in a sealed flask and equilibrium is established.
NH4CO2NH2(s) ⇌ 2 NH3(g) + CO2(g)
If the partial pressure of NH3(g) is 0.242 atm at equilibrium, determine the equilibrium constant, Kp.
- An aqueous mixture of hydrocyanic acid and ammonia has initial concentrations of 0.100 M HCN(aq) and 0.140 M NH3(aq). At equilibrium, the CN–(aq) concentration is 0.055 M. Calculate Kc for the reaction.
HCN(aq) + NH3(aq) ⇌ CN–(aq) + NH4+(aq)
- A 0.72-mol sample of PCl5 is put into a 1.00 L vessel and heated. At equilibrium, the vessel contains 0.40 mol of PCl3(g) and 0.40 mol of Cl2(g). Calculate the value of the equilibrium constant for the decomposition of PCl5 to PCl3 and Cl2 at this temperature.
- A sample of ammonium chloride was heated in a closed container:
NH4Cl(s) ⇌ NH3(g) + HCl(g)
At equilibrium, the pressure of NH3(g) was found to be 1.75 atm. What is the value of the equilibrium constant, KP, for the decomposition at this temperature?
- What is the pressure of BrCl in an equilibrium mixture of Cl2, Br2, and BrCl if the equilibrium pressure of Cl2 is 0.115 atm and the equilibrium pressure of Br2 is 0.450 atm?
Br2(g) + Cl2(g) ⇌ 2 BrCl(g) Kp = 4.7 × 10-2
- Calculate the number of moles of HI that are at equilibrium with 1.25 mol of H2 and 1.25 mol of I2 in a 5.00 L flask at 448 °C.
H2(g) + I2(g) ⇌ 2 HI(g) Kc = 50.2 at 448°C
- A student solved the following problem:
The problem was: For the following reaction at 600 °C:2 SO2(g) + O2(g) ⇌ 2 SO3(g) Kc = 4.32What are the equilibrium concentrations of all species in a mixture that was prepared with [SO3] = 0.500 M, [SO2] = 0 M, and [O2] = 0.350 M?
and found the equilibrium concentrations to be [SO2] = 0.590 M, [O2] = 0.0450 M, and [SO3] = 0.260 M. How could this student check the work without reworking the problem? - What are the equilibrium concentrations after a mixture in a sealed container, initially containing [H2O] = 1.00 M and [Cl2O] = 1.00 M, comes to equilibrium at 25°C?
H2O(g) + Cl2O(g) ⇌ 2 HOCl(g) Kc = 0.0900
- What are the concentrations of PCl5, PCl3, and Cl2 in an equilibrium mixture produced by the decomposition of a sample of pure PCl5 with [PCl5] = 2.00 M? Assume a constant temperature.
PCl5(g) ⇌ PCl3(g) + Cl2(g) Kc = 0.0211
- For the reaction: SO2(g) + NO2(g) ⇌ SO3(g) + NO(g), the equilibrium constant, Kc, is 18.0 at 1200 °C. If 1.0 mole of SO2 and 2.0 moles of NO2 are placed in a 2.0 L container, what concentration of SO3 will be present at equilibrium?
- If one starts with pure NO2(g) at a pressure of 0.500 atm, the total pressure inside the reaction vessel when 2 NO2(g) ⇌ 2 NO(g) + O2(g) reaches equilibrium is 0.674 atm. Calculate the equilibrium partial pressure of NO2.
- Graphite, which is a solid form of carbon, reacts with carbon dioxide, CO2, to form carbon monoxide, CO:
C(graphite, s) + CO2(g) ⇌ 2 CO(g)
At 727 °C, the equilibrium constant for this reaction, Kc, is 0.021. If 12.0 grams of graphite and 1.80 grams of CO2 are placed in a 2.00 liter reactor and heated to 727 °C, what is the concentration of CO at equilibrium?
- What is the minimum mass of CaCO3 required to establish equilibrium at a certain temperature in a 6.50 L container if the equilibrium constant (Kc) is 0.050 for the decomposition reaction of CaCO3 at that temperature?
CaCO3(s) ⇌ CaO(s) + CO2(g) Kc = 0.050
- In a 3.0 L vessel, the following equilibrium partial pressures are measured: N2 = 190. torr, H2 = 317 torr, and NH3 = 1.00 × 103 torr.
N2(g) + 3 H2(g) ⇌ 2 NH3(g)
How will the partial pressures of H2, N2, and NH3 change if H2 is removed from the system? Will they increase, decrease, or remain the same?
- The equilibrium constant (Kc) for this reaction is 5.0 at a given temperature:
CO(g) + H2O(g) ⇌ CO2(g) + H2(g)
- On analysis, an equilibrium mixture of the substances present at the given temperature was found to contain 0.20 mol of CO, 0.30 mol of water vapor, and 0.90 mol of H2 in a liter. How many moles of CO2 were there in the equilibrium mixture?
- Maintaining the same temperature, additional H2 was added to the system, and some water vapor was removed by drying. A new equilibrium mixture was thereby established containing 0.40 mol of CO, 0.30 mol of water vapor, and 1.2 mol of H2 in a liter. How many moles of CO2 were in the new equilibrium mixture? Compare this with the quantity in part (a), and discuss whether the second value is reasonable. Explain how it is possible for the water vapor concentration to be the same in the two equilibrium solutions even though some vapor was removed before the second equilibrium was established.
- Chlorine, Cl2, is a green and very toxic gas. The Cl2 molecule is reasonably stable at room temperature but at higher temperatures the bond between the two atoms will break forming two Cl atoms as described by the equation below.
Cl2(g) ⇌ 2 Cl(g) Kp = 1.3 × 10-4 (1100 °C)
- Write the equilibrium constant expression, Kp, for the dissociation of Cl2 molecules to Cl atoms.
- If the mixture of gases in the flask are at equilibrium, and the partial pressure of Cl2 is 0.500 atm (at 1100 °C), what is the partial pressure of the Cl atoms?
- When the temperature of the flask and contents decreases, the contents of the flask become darker green in color. Is the forward reaction endothermic or exothermic?
- If the volume of the flask containing the gas mixture from part (b) (at equilibrium and 1100 °C) is reduced by a factor of 2, determine Qp for the system immediately after the change, and predict which way the equilibrium will shift to reestablish equilibrium.
- Determine the partial pressures of the Cl2 and Cl atoms in the smaller flask (part (d)) after equilibrium has been re-established between the two species.
- When a soluble source of thiocyanate ions, SCN–(aq), is added to a solution containing Fe3+(aq), the solution turns from a pale yellow to a deep red color due to the formation of FeSCN2+(aq). The equilibrium established between the Fe3+(aq), SCN–(aq) and Fe(SCN)2+(aq) is described below:
Fe3+(aq, pale yellow) + SCN–(aq) ⇌ Fe(SCN)2+(aq, deep red) K = 5.4 × 104
- Which way would the reaction shift if Na(SCN)(aq) is added to an equilibrium mixture of Fe3+(aq), SCN–(aq), and Fe(SCN)2+(aq)?
- Which way would the reaction shift if Pb(NO3)2(aq) is added to an equilibrium mixture of Fe3+(aq), SCN–(aq), and Fe(SCN)2+(aq), given that lead(II) thiocyanate is insoluble?
- If enough water is added to an equilibrium mixture of the three ions so that the volume is effectively doubled, would the value of Q, the reaction quotient, increase, decrease or remain the same?
- If enough water is added to an equilibrium mixture of the three ions so that the initial volume is effectively doubled, which way does the reaction shift to reestablish equilibrium, if necessary?
- If the initial concentration of Fe(SCN)2+(aq) is 0.10 M (and the other two ions are 0.0 M), what are the concentrations of all three species when equilibrium is established?
Answers to Chemistry End of Section Exercises
- D
- Kp = 7.09 × 10-3
- Kc = 0.79
- Kc = 0.50
- Kp = 3.06
- PBrCl = 0.0493 atm
- 8.86 moles
- Calculate Q based on the calculated concentrations and see if it is equal to Kc. Because Q does equal 4.32, the system must be at equilibrium.
- [H2O] = 0.870 M, [Cl2O] = 0.870 M, [HOCl] = 0.260 M
- [PCl5] = 1.81, [PCl3] = 0.195 M, [Cl2] = 0.195 M
- 0.48 M
- 0.152 atm
Left-click here to watch Exercise 12 problem solving video.
- 0.016 M
- 32.5 grams
- Removing H2 gas from the system will disturb the equilibrium, the resulting QP value will be larger than Kp. This will cause the equilibrium to favor the reverse reaction until equilibrium is reestablished. After equilibrium is reestablished, the pressure of N2 will be higher and the pressure of the NH3 will be lower than prior to removing the hydrogen. The hydrogen pressure will go down quickly and then increase, but not to a pressure as high as before the removal so it will be lower.
- (a) 0.33 moles
(b) CO2 = 0.50 moles; Added H2 forms some water to compensate for the removal of water vapor and as a result of a shift to the left after H2 is added. - (a)
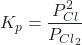
(b) 8.1 × 10-3 atm
(c) Taking away heat by cooling the flask results in more Cl2. This means heat must be on the reactant side: heat + Cl2(g) ⇌ 2 Cl(g); therefore, the reaction is endothermic.
(d) Qp = 2.62 × 10-4. Qp > Kp, the equilibrium will shift to produce more reactants.
(e) PCl2 = 1.00 atm, PCl = 0.0114 atm - (a) Towards products
(b) Towards reactants
(c) Q would increase
(d) Towards reactants
(e) [Fe3+] = [SCN–] = 0.0014 M, [Fe(SCN)2+] = 0.10 M
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

