D26.2 Amino Acids
Structure
Amino acids are amphiprotic because each amino acid molecule contains a carboxylic acid group that can donate a proton and an amine group that can accept a proton. Carboxylic acids are moderately acidic, many with Ka of ~10-5. Organic amines are somewhat basic, many with Kb of ~10-4. This combination creates an interesting situation, where an acid-base reaction is possible within a single amino acid molecule:

The carboxylic acid group, with Ka = 10-5, is a stronger acid than the protonated amine group, with Ka = Kw/Kb(amine) = 10-14/10-4 = 10-10. The amine group (Kb = 10-4) is a stronger base than the carboxylate anion (Kb = 10-9). The stronger acid and stronger base are on the left side of the above equation, so this reaction is product-favored at pH = ~7. Hence, at the pH of a typical living organism, the amino acid is a zwitterion (German for “double ion”), a species with no overall electrical charge but with separate parts that are positively and negatively charged.
The formation of a zwitterion is analogous to the acid-base reaction between methylamine (Kb = 4.4 × 10-4) and acetic acid (Ka = 1.8 × 10-5):
where the equilibrium favors products because:
Increasing the pH of an amino acid solution by adding hydroxide ions can remove the hydrogen ion from the -NH3+ group:
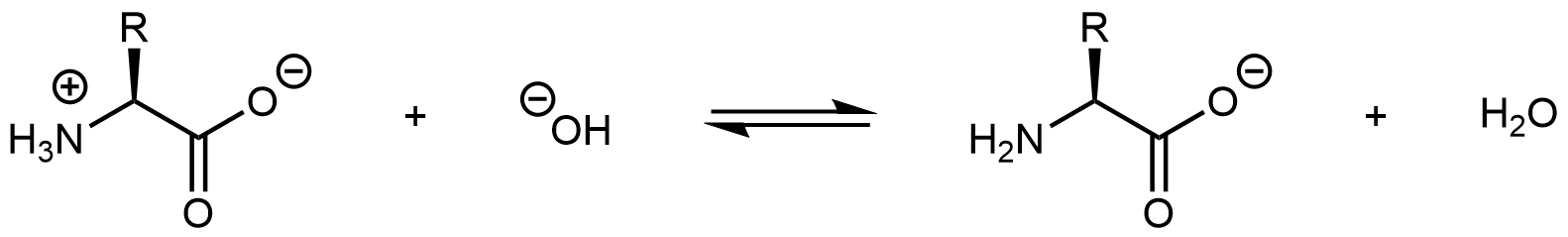
The product molecule is no longer a zwitterion. Instead, it is an anion with an overall charge of -1.
Similarly, decreasing the pH by adding strong acid to an amino acid solution protonates the -COO– part of the zwitterion:
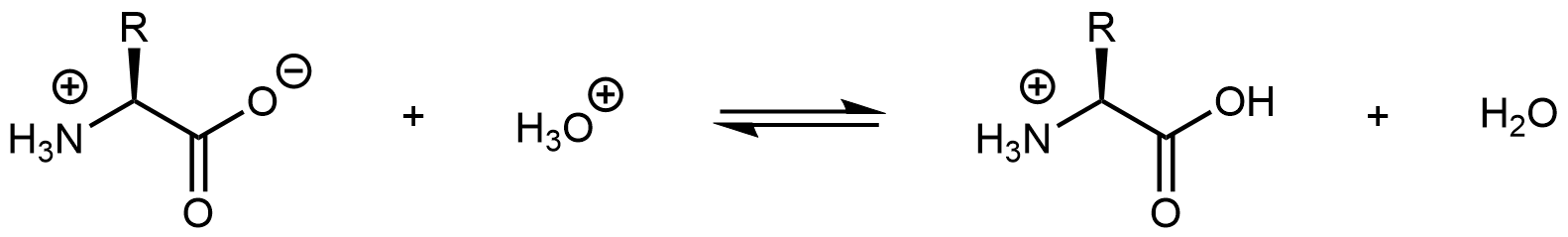
Again, the product molecule is not a zwitterion, but a cation with an overall charge of +1.
Classification
The twenty amino acids found in human proteins are often sorted into four groups—hydrophobic, polar but neutral, basic, and acidic—depending on the nature of their side chain “R” group.
The amine and carboxylic acid groups are present in all amino acids, hence they are not used to classify different amino acids. Moreover, the amine and carboxylic acid groups undergo condensation reactions to form amide linkages when amino acids polymerize to form proteins. Therefore, in a strand of protein, the side chains of each amino acid branch off the protein backbone and can influence protein 3D structure. Knowing the nature of these side chains is important for understanding protein structure.
Hydrophobic side chain
The nine amino acids that have hydrophobic side chains are shown below:
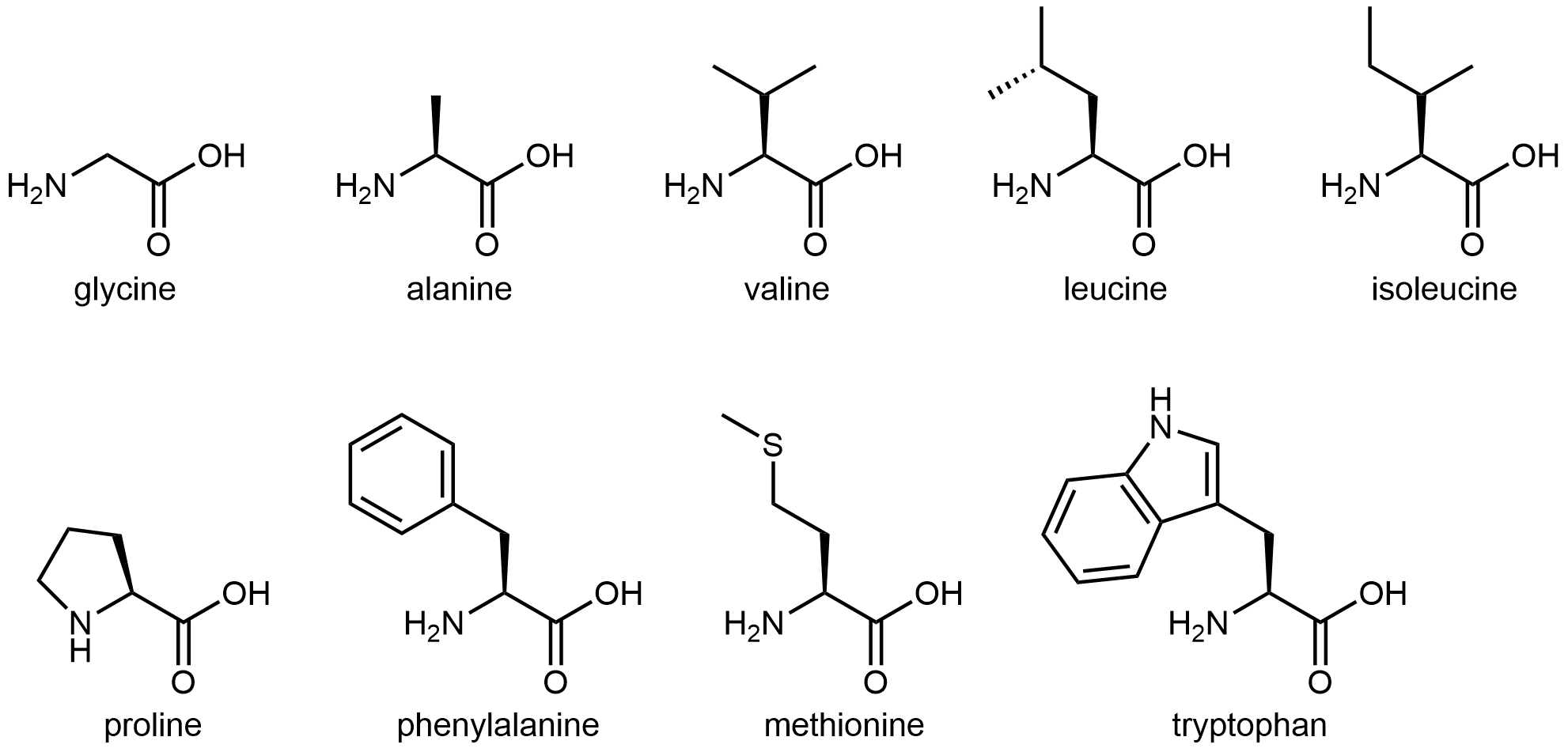
Seven of these amino acids have an entirely alkyl side chain (glycine’s side chain is simply an H atom, proline’s side chain forms a ring with its amine group), which can only interact with water molecules via London dispersion forces. These weak intermolecular interactions with water molecules make them hydrophobic.
Methionine’s side side contains a thioether group (similar to an ether, but with an S instead of an O), which is somewhat polar but cannot form hydrogen-bonding interactions with water molecules. Hence, methionine’s side chain is also classified as hydrophobic.
Tryptophan has a large side chain, composed mostly of carbon and hydrogen. The double ring structure is aromatic, meaning that the nitrogen-containing part of the side chain is more similar to an amide than an amine, and it is not basic. However, the N-H bond can have hydrogen-bonding interactions with water molecules, although it is insufficient to overcome the hydrophobic nature of the rest of the side chain. Tryptophan side chain, as a whole, is still classified as hydrophobic.
Polar side chain
The six amino acids shown in below have side chains that are polar, but neither acidic nor basic.
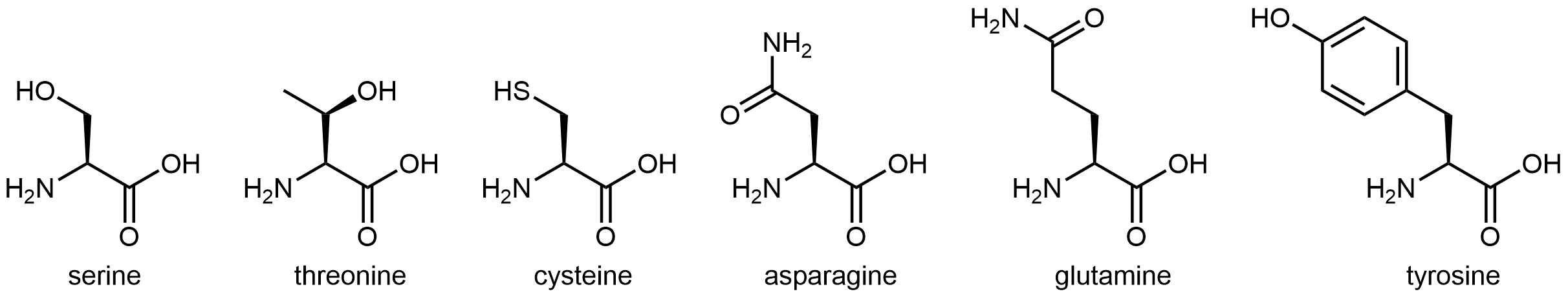
All of these amino acids have a side chain that contains a functional group with significant dipole moment. Five of them can also form strong hydrogen-bonding interactions with water molecules (containing either an alcohol group or an amide group). These characteristics make these side chains hydrophilic.
Tyrosine does have an aromatic ring in its side chain, but it is not as sizeable as the side chain in tryptophan. In addition, the -OH group has stronger hydrogen-bonding interactions with water molecules (it has O-H bond as well as lone pair on O atom) compared to tryptophan side chain. Overall, tyrosine side chain is more hydrophilic than tryptophan side chain.
Cysteine has a thiol (-SH) functional group. While it does not form strong hydrogen bonds with water molecules, it does impart a dipole moment to this relatively small side chain, making this side chain overall polar.
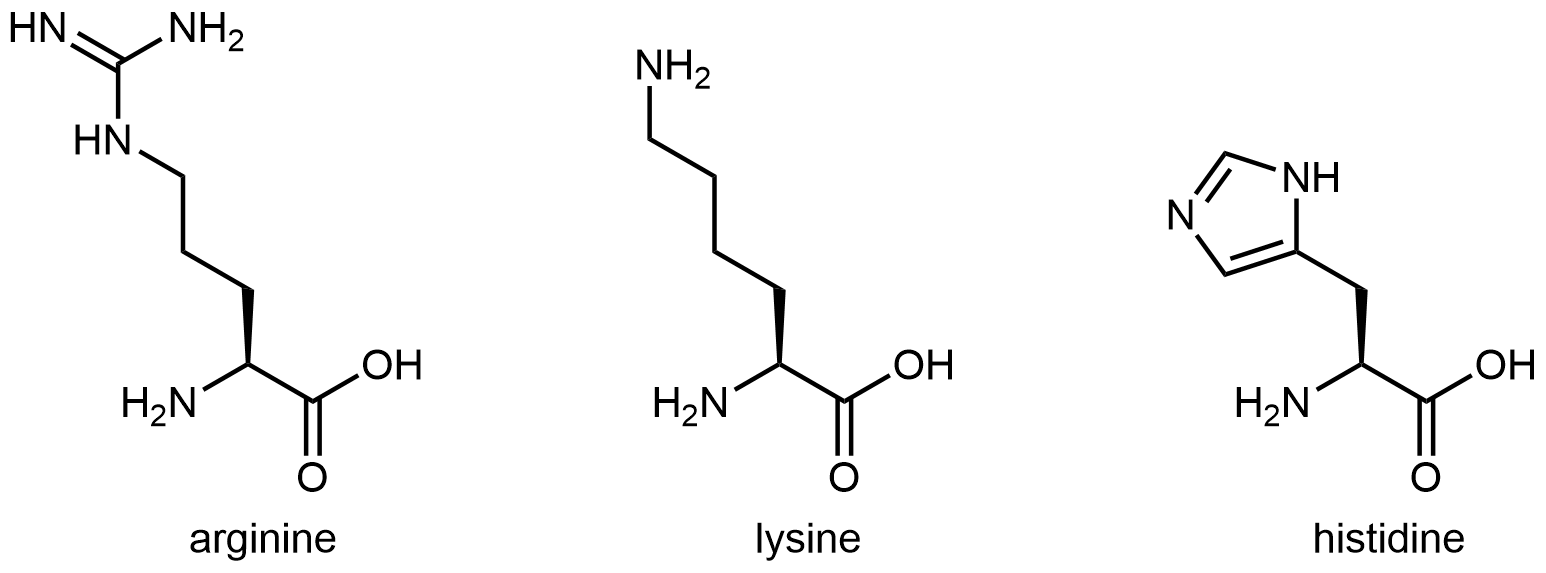
Basic side chain
The three amino acids below have side chains containing functional groups that are basic:
Activity: What Part of the Histidine Side Chain Is Most Basic?
The amine functional group at the end of the side chain in lysine imparts basic characteristics to that side chain. The functional group in arginine’s side chain is more complex, but similar to the analysis in the activity above for histidine, you can determine which nitrogen is more important in making the side chain basic by drawing and analyzing a set of resonance structures.
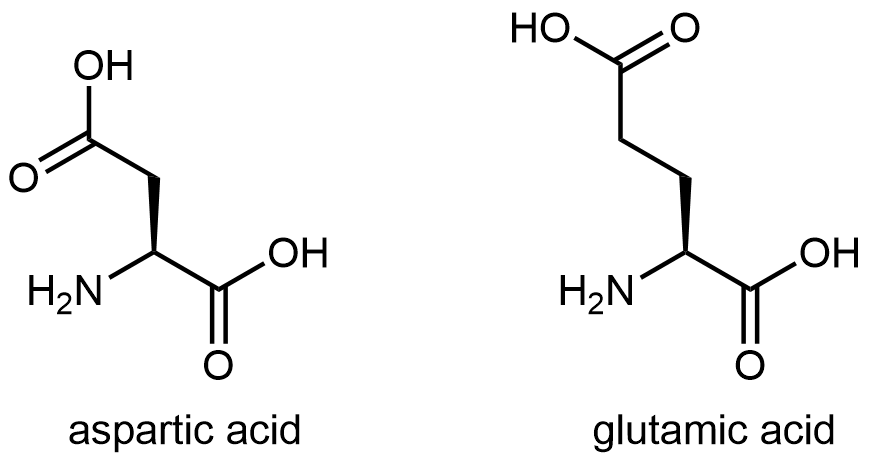
Acidic side chain
The figure below shows two amino acids that have an acidic side chain containing the carboxylic acid functional group:
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

