D6.3 MO Electron Configuration and Bond Order
Just as an atom has an electron configuration, so does a molecule. The molecular ground-state electron configuration can be obtained by filling electrons into MOs whose relative energies are shown in diagrams such as the one shown in Figure: MO Energy Levels, following rules similar to those for atomic electron configurations:
- Sum all the electrons from all the atoms that make up the molecule.
- Each MO can hold at most two electrons. (Two electrons occupying the same orbital must have opposite spin.)
- In the MO diagram the MOs are arranged in order of increasing energy.
- Add electrons to the lowest-energy MO first and then to successively higher-energy MOs (Aufbau principle).
- If two or more MOs have the same energy (are degenerate), apply Hund’s rule: assign one electron to each degenerate MO before pairing any electrons.
For the H2 molecule: there is one electron from each H atom, so there are two electrons in H2. There is only one lowest-energy MO, σ1s, therefore the two electrons, with opposite spin, will fill it. The ground state electron configuration of H2 is (σ1s)2.
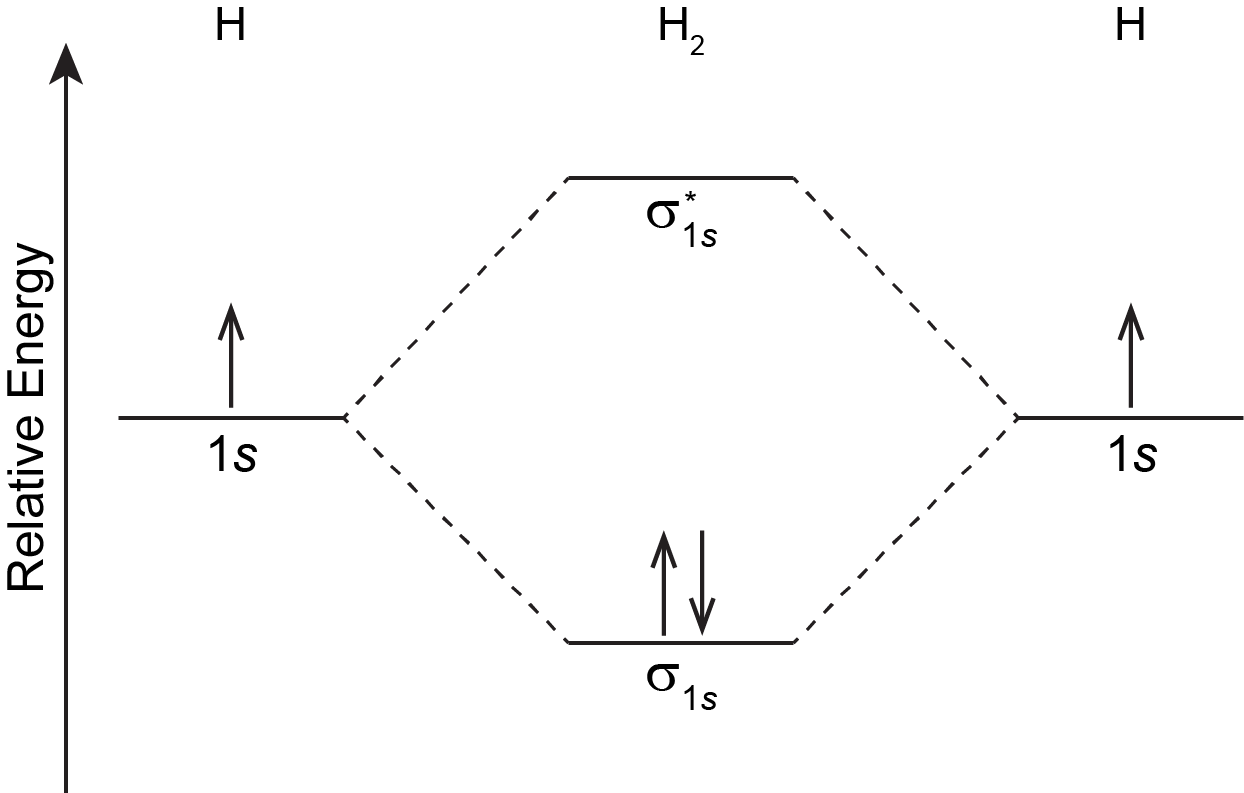
In Figure: MO Energy Levels in the previous section, and by convention in general, phases for occupied MOs are indicated by blue and red (when needed), while phases for unoccupied MOs are indicated by green and yellow. That is, σ1s is blue because it has two electrons and is all the same phase; σ*1s is green and yellow because there are no electrons in this MO and one part is opposite phase from the other.
Activity: Molecular Orbitals and Electromagnetic Radiation
The two electrons in the σ1s MO, which are shared between the two H atoms, form a covalent bond. The configuration (σ1s)2 indicates a single, sigma covalent bond. In general, a covalent bond involves overlap of two or more AOs that leads to increased electron densities close to atomic nuclei so that the electrons’ energies are lowered. Electrons are shared between pairs of atomic nuclei, or more widely among several nuclei within a molecule.
Bond Order
In some cases more than two electrons can be shared between two nuclei. This gives rise to the idea of bond order, which is the number of shared electron pairs between two atoms. Using a MO diagram, bond order can be defined as:
where nbonding e‾ is the number of electrons in bonding MOs and nantibonding e‾ is the number of electrons in antibonding MOs. There is a factor ½ because two shared electrons make one bond. In general, a greater number of shared electrons means a stronger bond, so the larger the bond order is, the stronger the bond is.
Exercise: Bond Order
Activity: Molecular Orbitals for He2
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

