D37.3 Homogeneous Catalysis
Reactions that are facilitated by catalysts can be divided into two major classes: homogeneous catalysis and heterogeneous catalysis. A homogeneous catalyst is present in the same phase as the reactants. We have already discussed several examples where catalyst and reactants are all in the same phase.
For example, we have previously described condensation reactions and hydrolysis reactions that occur in aqueous solutions. (For a condensation that produces water as the byproduct, hydrolysis is the reverse reaction.) We mentioned that condensation and hydrolysis can be catalyzed by strong acids. This is a homogeneous catalytic reaction because reactants, products, and catalyst are all in aqueous solution.
An example of ester hydrolysis is the acid-catalyzed decomposition of methyl acetate to form acetic acid and methanol. The reaction mechanism is shown below.
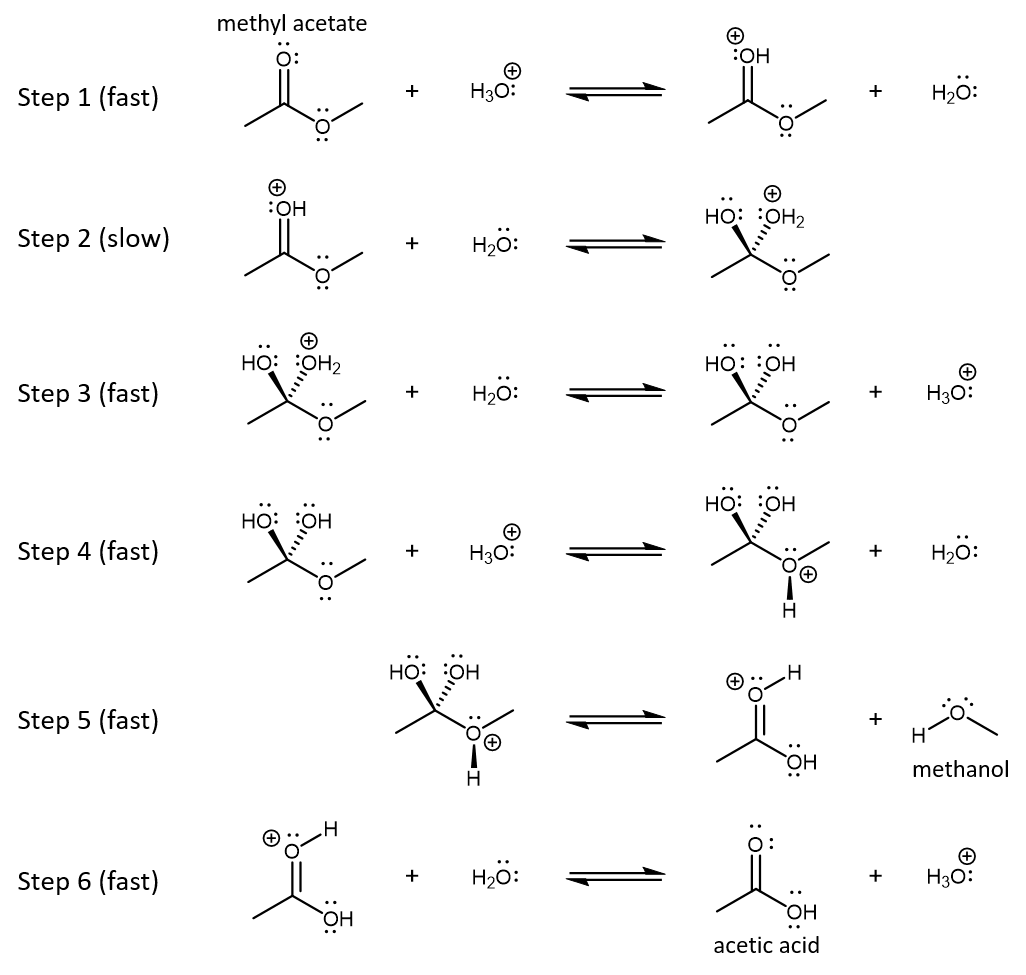
Activity: Analyzing a Reaction Mechanism
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

