D17.2 Energy, Temperature, and Heat
Thermal energy is kinetic energy associated with the random motion of atoms and molecules. When thermal energy is transferred into an object, its atoms and molecules move faster on average (higher KEaverage), the object’s temperature increases, and we say that the object is “hotter”. When thermal energy is transferred out of an object, its atoms and molecules move more slowly on average (lower KEaverage), the object’s temperature decreases, and we say that the object is “colder”.
Heating (or heat), represented by q, is the transfer of thermal energy between two bodies at different temperatures. Heat transfer of energy continues until both objects have reached the same temperature; that is, until thermal equilibrium has been reached.
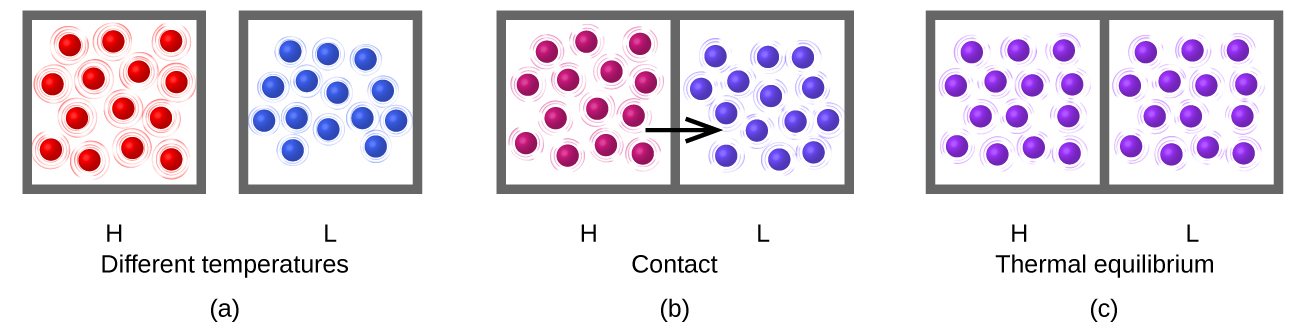
Consider what happens during the heat-transfer process: energy transfer occurs as a result of collisions between molecules. (The boxes drawn around the groups of molecules in the figure are to delineate which molecules are in which group, but the molecules contact each other.) On average the molecules in the hotter sample move faster than the molecules in the cooler sample, so on average a molecule from the hotter sample can transfer more energy than a molecule in the cooler sample. Thus, as long as the temperature is different, energy transfers from hotter to cooler.
After thermal equilibrium has been reached, energy transfer by molecular collisions continues to occur, but now the probability of energy transfer from sample H to sample L equals the probability of energy transfer from sample L to sample H. On the macroscopic scale there is no change in temperature of either sample, but on the atomic scale energy transfer continues at equal rates H→L and L→H. The atoms do not stop moving and transferring energy but the transfers are equal and opposite. This is a general characteristic of equilibrium.
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

