D35.4 Unimolecular Elementary Reactions
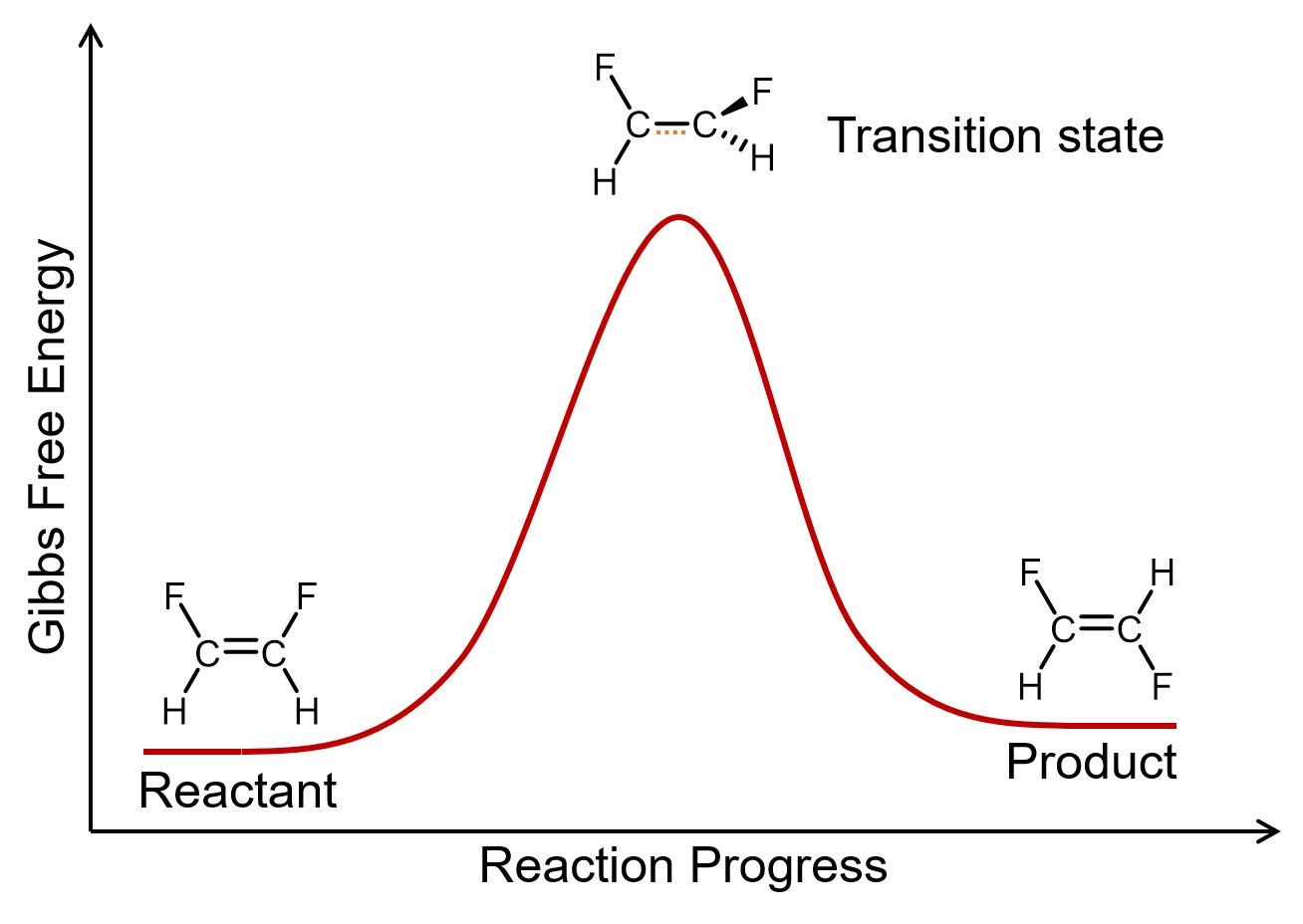
A unimolecular elementary reaction involves the rearrangement of a single reactant molecule to produce one or more product molecules. The cis–trans isomerization of 1,2-difluoroethene, step 1 in the mechanism for ozone decomposition, and many radioactive decay reactions, are all examples of unimolecular elementary reactions.
A general equation for a unimolecular elementary reaction is:
Suppose that a 1.0-L flask contains 0.0010 mol of reactant A. The concentration of A is 0.0010 mol/1.0 L = 0.0010 M. At a given temperature, a tiny fraction of the A molecules has enough energy to overcome the activation-energy barrier and react. If we add another 0.0010 mol of A to the flask, the number of A molecules doubles and the concentration of A doubles to 0.0020 M. If the temperature remains the same, the fraction of molecules that has enough energy to react remains the same, but now there are twice as many molecules so the number of molecules reacting (and the rate of reaction) doubles; that is, the rate of reaction is directly proportional to the concentration of A. Thus, the rate equation for a unimolecular reaction is:
If a reaction is known to be a unimolecular elementary reaction, its reaction rate is directly proportional to the concentration of the reactant and the reaction is overall first-order. We see here that because an elementary reaction describes exactly the reaction that is occurring, it is possible to determine the order of an elementary reaction solely by looking at the reaction equation. This is true for all elementary reactions.
Exercise: Unimolecular Reactions
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

