D36.1 Bimolecular Elementary Reactions
The collision and reaction of two molecules or atoms in an elementary reaction is a bimolecular elementary reaction. There are two general types of bimolecular elementary reactions. In one type, the two reactants are different:
The rate law for such a reaction is first-order in [A] and first-order in [B]:
In the other type of bimolecular elementary reaction, the two reactants are the same:
The rate law for such a reaction is second-order with respect to [A]:
If a reaction is known to be a bimolecular elementary reaction, then its rate law can be derived by considering how concentration of each reactant affects the number of collisions. This is shown in the figure below for the case of “A + B ⟶ products”. Assuming that the fraction of collisions that results in reaction is the same in all three cases, the rate of reaction doubles when the number (and concentration) of either reactant doubles. The rate law derived from number of collisions agrees with the rate law derived from the reaction equation. A bimolecular reaction is overall second-order.
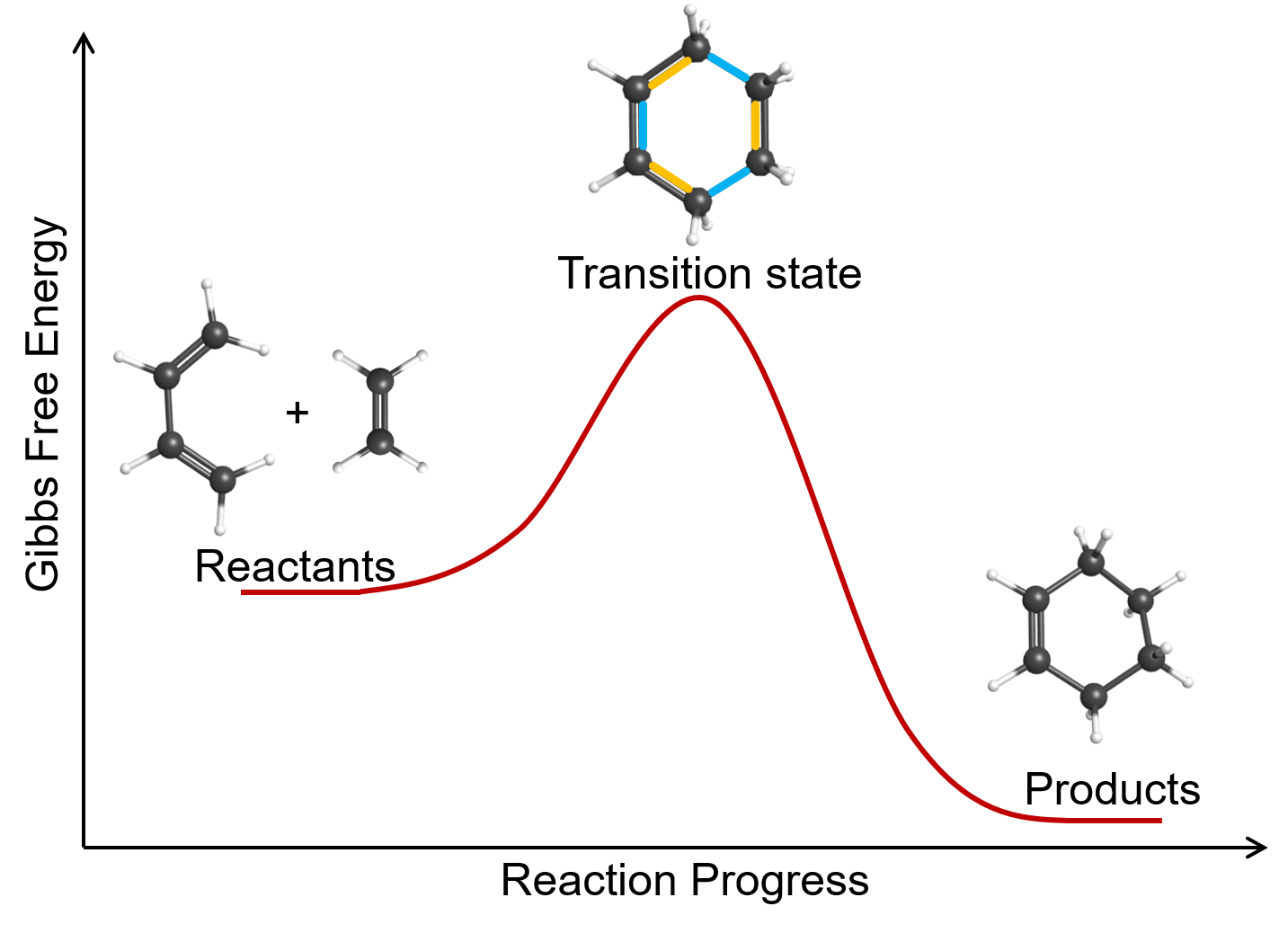
Some chemical reactions have mechanisms that consist of a single bimolecular elementary reaction. An example is the gas-phase reaction of 1,3-butadiene with ethene to form cyclohexene:
Another example of a bimolecular elementary reaction is step 2 in the ozone decomposition mechanism given earlier:
Exercise: Bimolecular Reactions
Exercise: Steric Factor
Please use this form to report any inconsistencies, errors, or other things you would like to change about this page. We appreciate your comments. 🙂

